|
The ability to image solid surfaces in a liquid medium makes atomic force microscopy (AFM) an attractive tool in the study of liquid-solid interfacial phenomena. One such area of research where AFM has proven useful is in the study of small particles and biological molecules that adsorb from an aqueous liquid onto a solid surface.
AFM provides magnifications great enough to resolve single, deep sub-micrometer particles at the surface, while the presence of the liquid keeps the adsorbed particles in their native, hydrated states. For these reasons, AFM is being widely used in the study of adsorption of colloidal particles, including polymer latexes, mineral colloids, and protein molecules.
Conventional Contact AFM Images
Conventional contact mode AFM images particles that are attached firmly to a surface, such as with covalent bonds. However, when colloidal particles adsorb to a solid, they associate non-covalently with the surface through electrostatic, van der Waals, or hydrophobic interactions, so they can have high lateral mobilities.
In contact mode AFM, the dragging motion of the probe tip exerts a lateral force on the surface that can push around loosely adsorbed particles, preventing the imaging of particles in their natural arrangement on the surface. This problem is avoided by imaging in liquid with TappingMode™ AFM.
TappingMode AFM Images
With this technique, patented by Veeco Instruments, the probe tip rapidly oscillates and taps the surface lightly at the bottom of each oscillation cycle. The intermittent contact eliminates lateral forces on the surface by the scanning tip, and allows imaging of adsorbed particles without moving them on the surface.
Figure 1 shows a liquid TappingMode image of positively-charged polymer latex particles (amidine latex) adsorbed to mica in water. The layer of adsorbed particles can be imaged repeatedly without any movement of the particles.

Figure 1. TappingMode in liquid image of positively charged polystyrene latex particles adsorbed to mica (in water). The average particle diameter is 120nm. 3μm scan
Figure 2 shows the same 3μm x 3μm area, imaged moments later in water with contact mode. The blurred streaks in the image suggest that the adsorbed particles do not remain stationary as the probe tip scans over them.

Figure 2. Contact mode image in water of the same area in Figure 1. 3μm scan.
Figure 3 shows a broader area of the sample (7μm x 7μm) imaged in water with TappingMode. The damage to the layer of adsorbed particles caused by the contact mode scan is clear. The adsorbed particles appear to have been pushed into clusters, mostly near the sides of the previously scanned region, and the bare mica substrate is exposed.
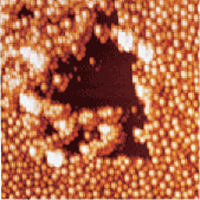
Figure 3. TappingMode image in water obtained after Figure 2. Damage to the adsorbed layer from the contact mode scan is seen where particles have been pushed into clusters, exposing bare areas of the mica substrate. 7μm scan.
Since the adsorbed particles are unaffected by the oscillating tip in TappingMode, it is possible to observe how the arrangement of particles at the surface is affected by system properties, such as the ionic strength of the surrounding liquid. This information is relevant to the processing of colloidal materials and the purification of protein products. With TappingMode in liquid, it is also possible to study how the layer of adsorbed particles grows with time, and to see how the structure of the adsorbed layer at the liquid-solid interface differs from the structure at air-solid interface.
Surface Modification of Probe Tips
For certain experimental systems, adsorbed particles may tend to adhere to the probe tip, even with TappingMode AFM. For example, positively charged latex particles will have an electrostatic attraction to the silicon nitride probe, which has a slightly negative surface charge in water. Such particles may become attached to the tip, resulting in artefacts in subsequent images. This problem is countered by modifying the probe surface with a silane coupling agent, 4-aminobutyldimethylmethoxy silane.
The monoethoxysilane deposits with sub-monolayer coverage on the probe, and the amino group on the attached silane confers a positive surface charge to the modified tip.
The positive charge on the tip inhibits adhesion of positively charged particles and greatly extends its lifetime for imaging such adsorbed colloids with TappingMode in water. This tip modification technique also works well when imaging dried adsorbed layers with TappingMode in air.
Summary
TappingMode imaging in liquids is an ideal experimental technique in the study of the adsorption of colloidal particles to solid surfaces. The elimination of lateral forces between the AFM tip, and the surface in TappingMode, allows particles at the liquid-solid interface to be imaged without changing their natural positions. This enables the study of the effects of solvent conditions on the structure of the adsorbed layer.
|