A visual examination of the bearing by Element Materials Technology revealed the loss of a significant amount of material from the surface. The exposed material was severely degraded, as apparent by the discoloration, loss of material integrity, and a network of perpendicular secondary cracking.
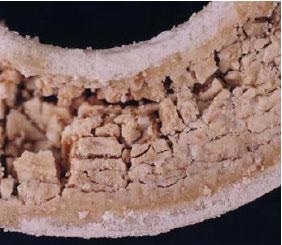
Figure 1. The bearing showed cracking and loss of material integrity, which was characteristic of severe chemical attack.
Core material from the bearing was analyzed using FTIR at Element Materials Technology, and the resulting spectrum was consistent with a polyacetal resin. However, due to the similarity in structure, polyacetal homopolymers and copolymers can not be differentiated spectrally, and a melting point determination is usually used to distinguish between these materials.
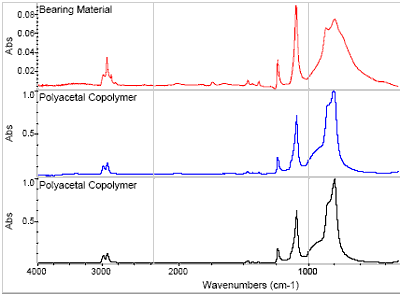
Figure 2. The FTIR results obtained on the bearing material were consistent with a polyacetal resin. However, spectral features were apparent, indicating molecular degradation.
Differential scanning calorimetry (DSC) was used to analyze the material and the resulting thermogram exhibited an endothermic transition characteristic of a melting point at 172°C. This melting point is indicative of a polyacetal homopolymer, and not a polyacetal copolymer as specified. While the two materials have similar structures, the subtle molecular variations result in performance differences, including thermal stability and chemical resistance. Chemical testing was performed on typical tomato-based products and the results indicated that such materials are moderately acidic, and have pH values ranging from 3 to 4.
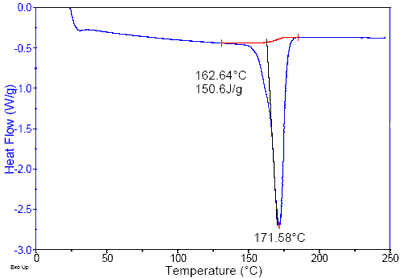
Figure 3. DSC testing showed the bearing material to have a melting point of 172°C, indicative of a polyacetal homopolymer.
Conclusion
It was the conclusion of the Element Materials Technology evaluation that the bearing failed through severe chemical attack, which produced molecular degradation. The bearing had been produced from a polyacetal homopolymer instead of the specified polyacetal copolymer. The dissimilarity in structure between these two materials, and the corresponding difference in chemical stability, particularly resistance to acidic materials, was sufficient to cause the observed failure. Polyacetal homopolymers are generally considered stable in contact with chemicals and solutions with pH values between 4 and 10, while polyacetal copolymers are recognized as being able to withstand contact with materials with pH values from 3 or 4 up to 14.
Fatigue and Fracture In Environment - Extended Version
Video Credit: Element Materials Technology

This information has been sourced, reviewed and adapted from materials provided by Element Materials Technology.
For more information on this source, please visit Element Materials Technology.