Photovoltaic cells and renewable power have advanced significantly as a result of legislative advantages and reduced system costs. This is mostly due to increased industrial utilization and a rise in power consumption as a result of both business and human operations. However, using solar and wind power is not dependable or consistent, causing electricity production to be erratic and intermittent, further testing the reliability of electrical installations.
Electric energy storage technology is proposed to address this unfavorable issue by smoothing the production of electricity generated.
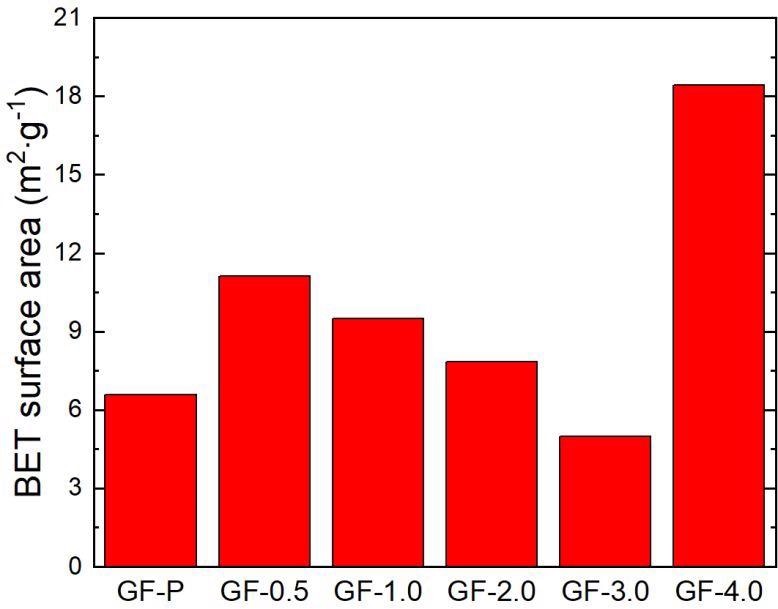
BET surface area of the carbon felt. Image Credit: Xia, L. et al., ACS Applied Energy Materials
What are Redox Flow Batteries?
Owing to the divergence of power and energy, a redox flow battery (RFB) is among the key systems for electricity and power storage. Redox-active materials (RAMs) immersed in solution are transferred into battery pyramids in conventional RFBs, and electrolytic changes proceed on the electrodes while the cell bundles run.
This indicates that RAMs and electrodes are both required for RFBs. Organic RAMs have received a lot of focus in recent years because of their possible low cost, "greenness," and characteristic adjustability. Currently, the majority of organic RAM research focuses on mechanism studies, chemical synthesis, and production.
Advantages of Redox Flow Batteries
One major benefit of redox flow batteries is that they are quite inexpensive. For a particular battery capacity, the exchanger has the same dimensions but the endurance power may be increased from four hours to more than 12 hours, merely by introducing bigger plastics storage facilities to carry additional solutions.
RFBs have various benefits, including the ability to be placed modularized and to autonomously vary the outgoing power generation capability by altering the number and size of units in a stack and the quantity of solution, respectively. They also have several future benefits, such as easy scalability (potential proportional to the storage tank, whereas output power roughly equivalent to PEM total area), no negative effects of overcharging, very low self–discharge, minimal rates for a substantial system as opposed to batteries, and long lifetime.
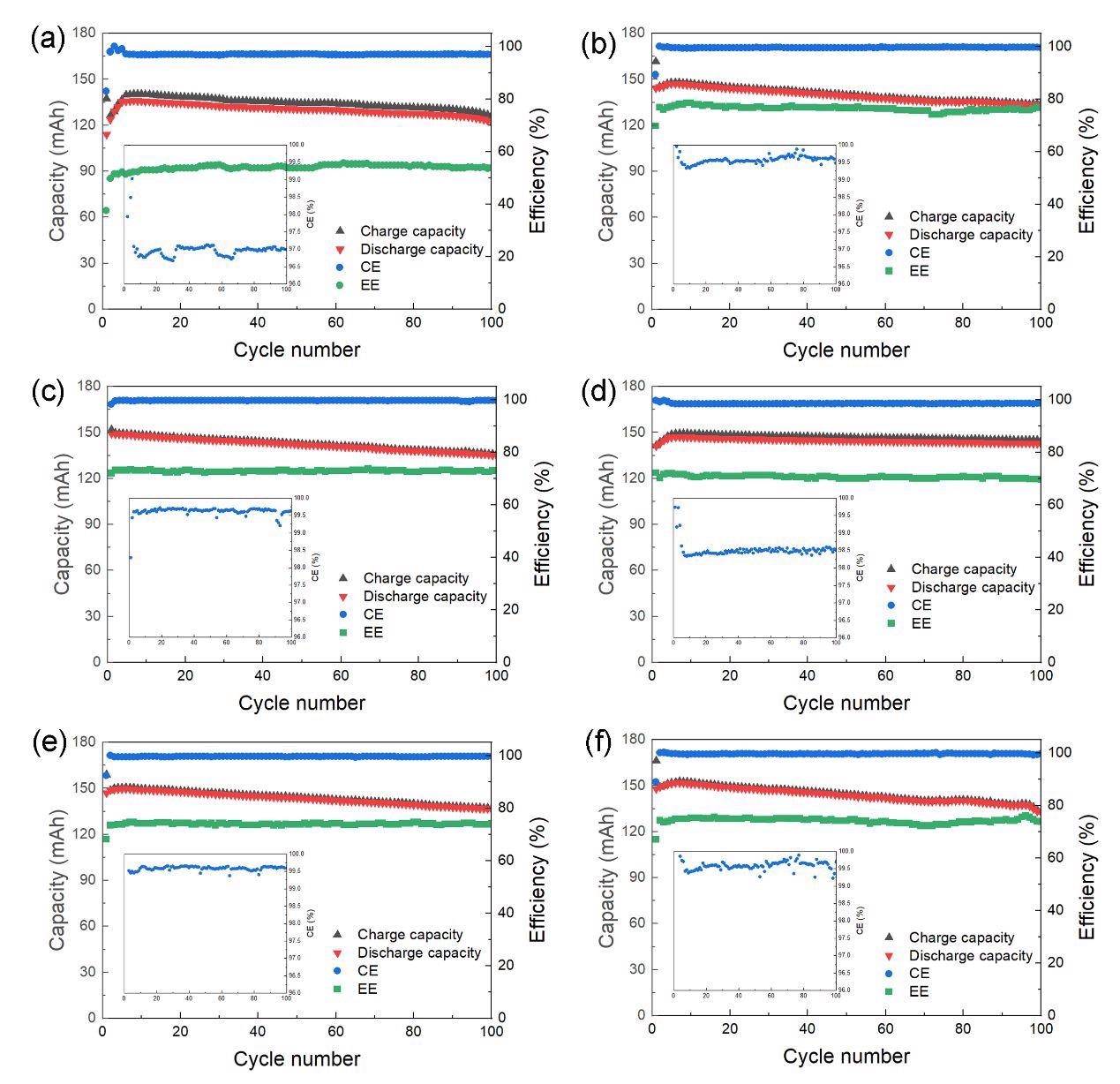
Battery performance of the quinone flow batteries by using pristine carbon felt (a) and thermally treated carbon felt at the heating time of 0.5 h (b), 1.0 h (c), 2.0 h (d), 3.0 h (e) and 4.0 h (f). All batteries were tested under 100 cycles at 100 mA·cm-2. Insets are corresponding enlarged pictures of corresponding CE. Image Credit: Xia, L. et al., ACS Applied Energy Materials
Limitations of Redox Flow Batteries
Although these features may seem to be ideal for large-scale industrialization, a few major factors hinder its thorough commercialization. The downsides of battery systems include the use of costly chemicals that are also abrasive or poisonous, as well as the reasonably high stability of operating cost and the parasitic (on-site) load required to operate the pumps.
The most well-known RFBs use aqueous electrolytes; nevertheless, the electrochemical window of water restricts the possibilities that these flow batteries may produce. Unfortunately, there are fewer electrodes or electrodes designed related to organic RFBs. Electrode manufacturing is thought to be necessary for biological RFBs as well. This is because the immaculate or untreated electrode is not ideal or suited for direct usage.
Introduction to Anthraquinones
Anthraquinones have a similar structure to anthracene. These chemicals give plants color and are commonly used as natural colorants. They are also used as laxatives and have antibacterial and antiviral properties. It is a yellowish and pure crystalline substance that is soluble in heated organic solvents but not in water. Anthraquinone acts as a redox mediator. The method of the process might entail a single electron transfer (SET). Anthraquinone is utilized as a solvent in flow batteries, which are capable of long-term power retention.
Thermal treatment, a common and easy approach, is employed in this study to activate a PAN-based carbon felt used in well-established 2,6-DHAQ/K4Fe(CN)6 RFBs.
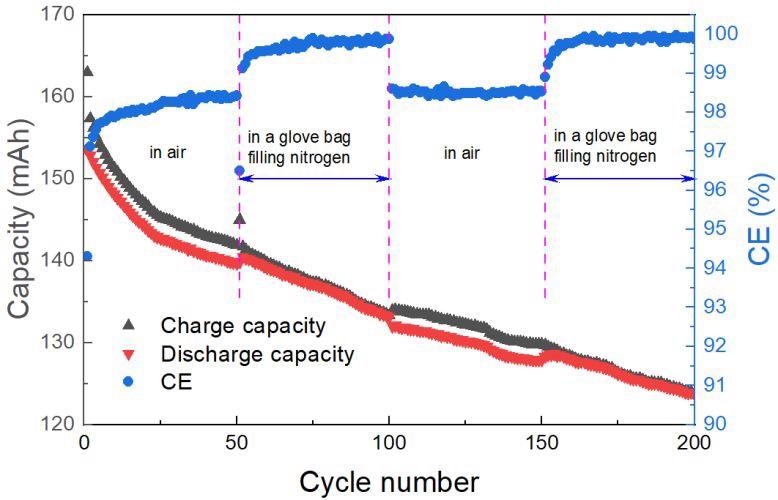
The RFB with CF-2.0 was examined under air during 1st ~50th and 101st ~150th cycles, and in a glove bag filled with nitrogen during 51st ~100th and 151st ~200th cycles. Image Credit: Xia, L. et al., ACS Applied Energy Materials
Research Findings
It was discovered that 600 °C was the optimum temperature for the carbon felt to promote air oxidizing without breaking the basic structure through combustion. After processing, the surface morphology of the carbon felt rose in general, providing more reactive groups.
As a result, the thermal degradation carbon felt has a higher current than the clean one. Importantly, when the pre-treatment was done, the Coulombic efficiency (CE) rose somewhat from 97 to 99 percent. Furthermore, as compared to CF-P, which has a fuel efficiency (EE) of 54%, the heated carbon felt had an EE of >70%. The battery also displayed superior durability after 200 cycles, with a capacity retention of 94.42 percent.
In short, PAN-based carbon felt had been thermally energized at 600 °C. The process demonstrates that by modifying electrodes, it is possible to increase the lifetime of quinone-based RFBs.
Further Reading
Xia, L. et al., 2022. Enhancing the Cycling Stability of Anthraquinone-Based Redox Flow Batteries by Using Thermally Oxidized Carbon Felt. ACS Applied Energy Materials. Available at: https://pubs.acs.org/doi/10.1021/acsaem.1c03507
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.