Silver products were used by the ancient Greeks to cure ulcers, encourage the healing of wounds, and preserve water and food. The growing amount and prevalence of antibiotic-resistant bacteria has reignited research into silver's antibacterial activities.
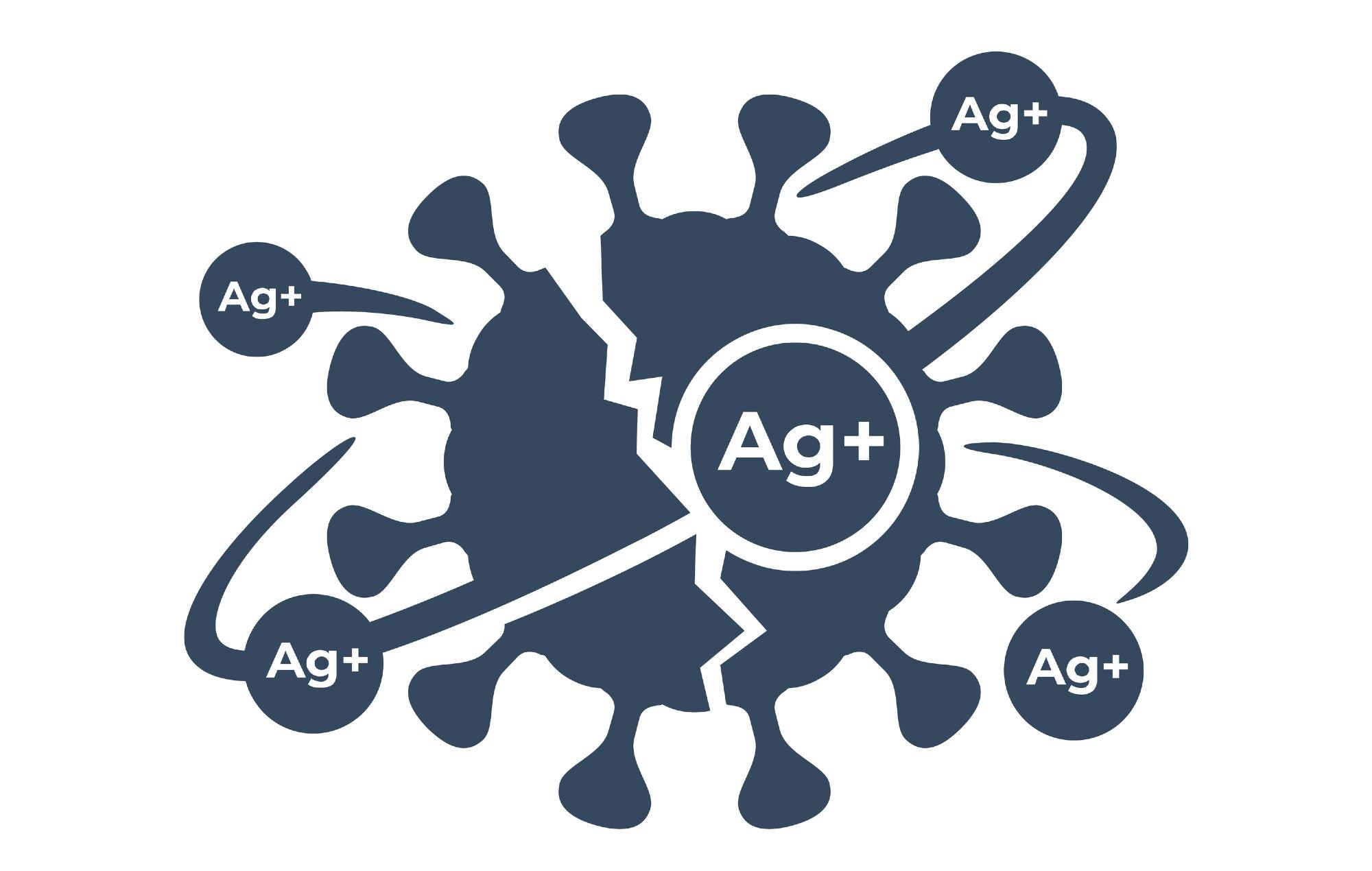
Image Credit: Dmitry Kovalchuk/Shutterstock.com
Silver Toxicity in Humans
There are few indications of silver toxicity in the human body, and clinical treatment poses only a minor risk. Silver is taken into the human body and circulates as a protein complex, which is then excreted by the kidneys and liver. The health concerns related to chronic silver ion absorption seem to be minimal.
Argyria and argyrosis, which are characterized by a permanent accumulation of silver selenide and silver sulfide precipitates in the skin and eyes, respectively, are the most prevalent visible abnormalities associated with extended exposure to silver compounds.
The damaged region becomes bluish-gray and gets worse when exposed to sunshine. Argyria and argyrosis, on the other hand, are not life-threatening and are not linked to permanent tissue damage. There are various suggested silver exposure limits and standards, although the levels vary depending on the reference body issuing the advice.
The World Health Organization determined that a lifetime silver consumption of roughly 10 g is the human no discernible harmful impact threshold.
Antibacterial Property of Silver
Silver quickly interacts with sulfhydryl groups on microbe surfaces by substituting hydrogen atoms, resulting in the creation of the S–Ag link. This fully shuts off respiration and electron transmission, making it difficult to initiate effective rescue processes. Furthermore, preventing respiration and electronic transfer leads the proton gradient to collapse, culminating in membrane de-energization and cell death.
As a by-product of cell metabolism, all aerobic organisms create reactive oxygen species (ROS). ROS are extremely harmful short-lived active oxidants that cause harm to enzymes, DNA, RNA, and fats. Recent research has shown that Ag treatment increases the generation of reactive oxygen species (ROS), which improves antibacterial activity.
More from AZoM: Thermal Properties of Chalcogenide Glass After Addition of Silver
Antioxidative enzymes rely on thiol groups for their function, which is hindered by silver ions. As a result, these enzymes are unlikely to be able to detoxify the ROS produced. Furthermore, bacteria grown anaerobically are generally less vulnerable to Ag ions, perhaps reflecting the effect of ROS generation on silver's antibacterial action.
Application of Antibacterial Silver
Silver's wide antibacterial properties have made it useful in a variety of medical applications, including containers, microscopic films, medical clothes, bedding and pillows, cosmetics, surgical equipment, wounds and burns treatment, biomedical devices, and a range of personal care products. Some of the detailed medical applications of silver are:
Sensors
The characteristics of the peptide and silver contact, as well as the influence of the peptide on the creation of silver nanoparticles, have been the focus of recent research on peptide capped silver nanoparticles for chromogenic sensors. Furthermore, silver nanoparticle-based fluorescence sensors have high efficiency and can exceed detection limitations.
Optical Probes
Raman scattering and metal-enhanced luminescence both employ silver nanoparticles as probes. Silver nanoparticles have greater benefits for probing than other noble metal nanoparticles, such as better attenuation coefficients, finer absorption bands, and strong field amplification.
Antimicrobial Agents
Fabrics, food preservation containers, refrigerator panels, and home healthcare devices all include silver nanoparticles and are the most extensively utilized sterilizing nanomaterial in consumable and medicinal products. The antimicrobial activity of silver nanoparticles is owing to the nanoparticles' persistent release of free silver ions.
Catalysts
For biocides like pigments, as well as chemical agents like toluene, silver nanoparticles have been shown to exhibit catalytic reactive characteristics. The catalytic characteristics of nanoparticles are influenced by their chemical environment. It's also worth noting that sophisticated catalysis is accomplished by the adsorption of reactant species on the catalytic substrate. The catalytic activity is frequently lowered when monomers, complex compounds, or lubricants are added as stabilizers or to avoid the convergence of nanoparticles due to lesser adsorption capacity. In particular, silver nanoparticles are utilized as catalysts in chemical reactions involving titanium dioxide.
Conclusion
Although the antibacterial impact of silver ions is not fully understood, it is believed that they interact with cell membranes, impeding electron transport and the proton motive force, resulting in improved antibacterial activity. Silver may be used for a variety of medicinal purposes. Future research should focus on the actual cause of silver toxicity and resistance.
A thorough understanding of all of these aspects may help to enhance the numerous uses of silver (such as antimicrobials, bioremediation, and nanomaterials) as well as better estimate the hazards to human health and the environment.
References
Mijnendonckx, K., Leys, N., Mahillon, J., Silver, S., & Van Houdt, R. (2013). Antimicrobial silver: uses, toxicity, and potential for resistance. Biometals, 26(4), 609-621. https://pubmed.ncbi.nlm.nih.gov/23771576/
Alexander, J. W. (2009). History of the medical use of silver. Surgical infections, 10(3), 289-292. https://pubmed.ncbi.nlm.nih.gov/19566416/
Edwards‐Jones, V. (2009). The benefits of silver in hygiene, personal care, and healthcare. Letters in applied microbiology, 49(2), 147-152. https://sfamjournals.onlinelibrary.wiley.com/doi/full/10.1111/j.1472-765X.2009.02648.x
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.