| A new type of ‘smart’ morphing polymer could shape the future of heart valves and other medical devices. Chemomechanical polymers, developed by Professor Hans-Jorg Schneider and his team at the University of Saarland, Germany, have greatly improved functionality compared to existing expanding / contracting materials used to perform biomedical functions, and could be used in applications such as actuators, implants, drug release systems and drug screening. Drug Delivery Systems Currently Available Drug Delivery Systems Previous materials used in these types of medical implants and systems have relied on one of two methods. Passive delivery systems use cyclodextrin as container or host molecules to liberate ‘trapped’ drugs with only one variable allowed, such as time. The second method relies on the reception of signals in abnormal conditions, such as changes in pH, or unusual concentrations of substances such as metal ions or metabolised drugs. These signals are then transmitted to additional electromechanical devices that are powered by implanted batteries. Automatic drug Delivery Systems The big breakthrough for the polymer hydrogels developed by Schneider is that they operate fully automatically, without the need for additional sensors, transducers, switches or pumps. Crucially, they also operate without an additional power supply - in contrast to existing techniques. Applications using the polymer are simplified due to the lack of moving parts, meaning easier maintenance and the development of more robust systems. It also allows miniaturisation, including use as thin film coatings on drugs. Chemomechanical Polymer Drug Delivery Systems How Automatic Drug Delivery Systems Work This direct response to different compounds or pH levels has been achieved by Schneider using a principle that rests upon the implementation of chemical recognition sites into the polymer. When changes (such as alterations in pH or compound concentration) occur in the solution surrounding the implant, the polymer can automatically open its gates to uptake (or release) a ligand from (or into) the surrounding aqueous solution. This has the effect of swelling or contracting the polymer. The polymers perform in a similar way to natural vessels or muscles in that they can perform macroscopic, reversible movements by the selective formation of supramolecular complexes. Different receptors can be applied to the polymer to program it to respond to alterations in different chemicals. ‘The system’s major principle of responding selectively to the presence or absence of a particular compound is completely novel,’ says Schneider. ‘In the area of chemically-induced drug release systems pH-dependent systems have been reported, but our polymer shows the mechanical effects at pH values both above and below the physiological pH.’ How Fast The Chemomechanical Polymers Can React to Changes These dimensional changes are completely reversible and relatively fast. A 50% expansion was seen in only a few minutes after exposure to the compound adenosine 5’-monophosphate (APM), figure 1, but Schneider anticipates that much faster rates of expansion could be achieved in the future. | 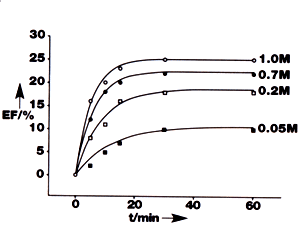
| | Figure 1. The expansion factor of the chemomechanical polymer as a function of time in response to different concentration of adenosine 5’-monophosphate at pH 6.8 compared with a 0.05M sodium phosphate buffer. | How The Chemomechanical Polymers Change Shape The polymer varies its shape when the pH changes, figure 2, since at neutral pH (7.0) the polyamines within the polymer are only partially extended and therefore more closely packed than at lower pH values. When positively charged nitrogen atoms repel each other in and around the chains, it causes the polymer to expand. At high pH values amines break free, forming extended chains with less hydrogen bonding interactions, causing the material to expand. | 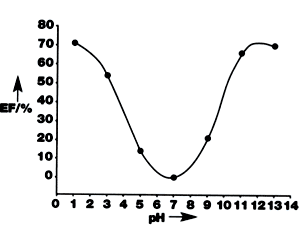
| | Figure 2. The expansion of the chemomechanical polymer as a function of changing pH. | Synthesis of The Chemomechanical Polymers One form of the polymer was synthesised by reacting poly(methyl methacrylate) with diethylenetriamine and long-chain alkyl amines. ‘We chose this blend of flexible polymer because it allows us to introduce many different receptor functions, enabling us to use the principle for a large variety of compounds,’ says Schneider. Peptides have recently been shown to produce sizeable dimension changes at well below millimolar concentrations. Applications Cardiovascular Implants Promising applications include forming actuators such as valves and switches from the polymer, which could be used as implants for blood vessels. The vessel could be widened or narrowed to regulate the blood flow, again using endogenous chemical stimuli control. The valves would either be implanted into blood heart vessels, or artificial muscle implants could be formed. Drug Screening Drug screening from combinatorial libraries and natural compound pools could also be supported using chemomechanical polymers in cantilever-type sensor array systems. Drug Release Systems But the most encouraging area, and the application that Schneider expects to hit the marketplace first, is that of drug release systems. A drug contained inside a microcapsule coated with the chemomechanical polymer can be released by external stimulation by substances present in malign tissues or blood, such as in tumour tissue, or by differences in pH. The principal could also be reversed to remove toxic compounds from the body. Summary So what is next for these smart materials? Following their success with peptides, the team intends to develop the polymers further using natural chitosan networks. And after drug release products are developed, sensor systems for drug screening are tipped to be next in line for commercial exploitation. |