Introduction Sialon ceramics (solid solution of Si3N4, mainly α-sialon (α’) and β-sialon (β’)) exhibit great potential for engineering applications because of their excellent mechanical properties, such as high hardness, toughness and strength [1]. Typically, sialons are prepared by firing powder compacts of Si3N4, AlN, Al2O3 and some oxide sintering additives at high temperatures via liquid phase sintering. Both the expensive starting powders and the complex manufacturing process (compared to most oxide ceramics) have limited the practical application of sialon ceramics. However, slag is a waste product from metallurgical plants and is produced on a daily basis in large quantities. Therefore, there are economical and ecological advantages for the reuse of this wasteful slag. Although the exact compositions of slag may vary in different plants, the basic composition of slag from a blast furnace contains CaO, MgO, SiO2 and Al2O3. There have been previous attempts to produce cement and glass out of slag [2, 3]. However, little literature is available on the use of slag to synthesize advanced ceramics, although its composition can be easily matched to that of the sialon ceramics if extra nitrogen is incorporated. In the present paper, we will summarize our recent work [4, 5] on preparation of α-sialon powders and bulk materials from slag-based starting materials. Experimental According to the slag composition (see Table 1, from Baosteel Company, Shanghai, China) and the Ca-Si-Al-O-N phase relationship, the nominal composition of α-sialon was designed as Ca0.71Mg0.23Si9.18Al2.82O0.94N15.06. In addition to the slag powders, the starting powders also include Si (Si>98 wt%, 30 μm), Al (Al>98 wt%, 25 μm). Small amounts of Si3N4 and AlN powders were used as seeds and dilution for the self-propagating High-temperature Synthesis (SHS) process and were counted into the nominal composition of α-sialon. The mixed powder was put into a graphite crucible and covered with a top layer of titanium powder. A tungsten-heating coil was connected to ignite the Ti powder, which then induced the spontaneous SHS process. The whole process was held in a sealed cabin with a high pressure of N2. The resulting powders were ball milled for 48 h and then sintered directly by hot-pressing (HP) at 1700ºC for 1 h and pressure-less sintering (PLS) at 1800ºC for 2 h in a graphite furnace. Table 1. Chemical composition of the slag from Bao Steel Company. | | | wt% | 39.76 | 34.48 | 14.04 | 9.18 | .52 | .09 | .83 | 1.36 | .25 | .31 | .58 | .27 | The phase analysis and morphology observation of powders were carried out by X-ray and SEM equipped with EDS. Bulk densities of the samples were measured according to Archimedes principle. Hardness and indentation fracture toughness on the hot-pressed samples were measured using a Vickers diamond indenter under a load of 98 N. Bending strength was measured at room temperature with an INSTRON-1195 using the three-point bending test with a span length of 30 mm and a cross-head speed of 0.5 mm/min. The erosion tests were carried out in a gas-blast type erosion test rig, which has previously been described in detail by Yu Zhang [6]. Results and Discussion Powder Synthesis The SHS-synthesised α-sialon powders exhibit non-uniform grain morphologies, as shown in Figure 1. Some grains have a large elongated shape and the others show fine equiaxed morphology. This can be attributed to the complexity of the reactions taken place in a very short time. After ball milling for 48 hours, the particle size of the SHS synthesized α-sialon powders was reduced to around 1μm. The XRD and EDS (Figure 2) results indicated that the synthesized powder were (Ca, Mg)- α-sialon. | 

| | Figure 1. SEM images of slag-sialon powder by SHS (a) original form SHS (b)after ball-milled for 48 h. | | 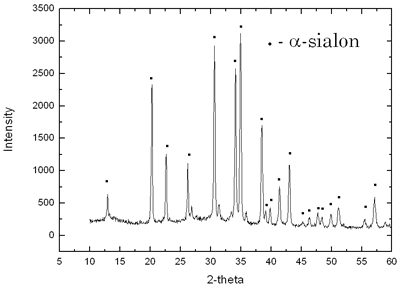
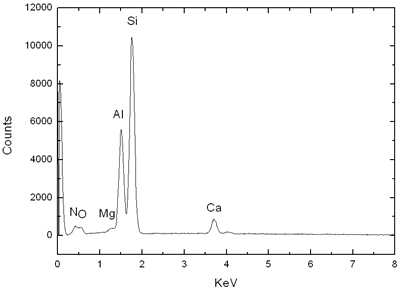
| | | Figure 2. XRD and EDS of SHS α–sialon powder. | | | | | Mechanical Properties The density and mechanical properties of the HP and PLS sintered samples are presented in Table 2. The theoretical density of slag α-sialon is 3.19g/cm3, obtained by measuring the density of the slag α-sialon powder. Hence, the HP sample attained a bulk density of > 99% and PLS samples achieved a bulk density of 96% of the true density. It is also seen in Table 2 that the strength of the HP sample reached 599 MPa and the hardness and toughness of PLS sample were 15.53 GPa and 4.72 MPam1/2. Table 2. Properties of HP- and PLS-slag-sialon samples [5]. | | | PLS | 3.07 | 96% | 15.53 | 4.72 | 360 | | HP | 3.17 | 99% | 16.02 | 4.48 | 599 | These properties are very close to that of normal α-sialon ceramics prepared using very expensive chemical starting powders. Although the HP sample is dense, the hardness is lower than normal HP α-sialons (generally above 18 GPa). This might be attributed to the presence of more glass at grain boundaries, which is soft and brittle. The lower strength of PLS sample is related to its low density and weak bonding between grains. Erosion Resistance The slag-derived α-sialons, densified by either pressureless sintering or hot-pressing, demonstrated excellent erosion resistance. Figure 3 shows the cumulative weight loss due to erosion of the ceramic samples as a function of the amount of SiC erodent impinging on the material surface at a 90 degree angle. For comparison, the erosion curves for a commercial silicon nitride (Si3N4), a commercial reaction bonded silicon carbide (RBSiC) and a Ca-α-sialon ceramic prepared using high purity powders by pressureless sintering at 1800°C for 2 h were also included in Figure 3. | 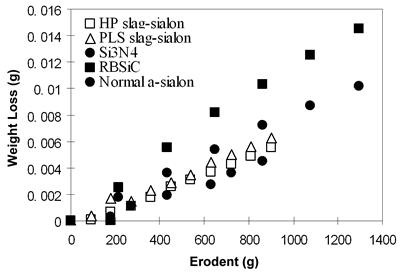
| | Figure 3. Figure 8 Cumulative weight loss of different materials resulted from SiC erosion. | It is seen that both the HP and PLS sintered slag-sialon materials showed similar erosion resistance as the Ca α-sialon ceramic prepared from high purity powders, but demonstrated better performance than the two commercial anti-wear ceramics. The microstructure of the eroded surface revealed the major material removal mechanism to be grain ejection (Figure 4). At 90 degree impact, the kinetic energy of the impinging particles mainly contributes to repeated impact, responsible for initiating and propagating grain boundary microcracks [7]. The superior anti-erosion performance of the Ca α-sialons is attributed to their combined high toughness and hardness. Thus, potential for the slag-sialons to be used as low-cost anti-erosion/wear materials has been demonstrated. | 
| | Figure 4. SEM microstructure of the HP slag-sialon sample after the erosion. | Industrial Applications Preliminary development for industrial applications has been explored, as shown in Figure 5. A Chinese company has produced ceramic bearings from the slag-sialons and their performance is under assessment. Ceramic tiles made from the slag-sialons have been tested at a Chinese industrial plant. In the test, six slag-derived sialon ceramic tiles were glued by epoxy on the surface of a steel plate which was then installed on the surface of a steel funnel that was exposed to a flowing stream of wet slag sands. Although some of the tiles were lost at various stages of the test due to debonding of the glue, the remaining ceramic tile was almost intact while the steel plate was badly eroded/corroded after two months of service. | 

| | Figure 5. Potential applications for slag-sialon ceramics (a) bearing ball and (b) tile. | Conclusions The SHS technique has greater potential for producing low-cost sialon materials. The α-sialon powder synthesized from slag displays properties suitable for the preparation of ceramic products. The as-sintered slag α-sialon samples showed good mechanical properties and excellent anti-erosion/wear properties. Because of the low cost of this process and the starting materials, future applications of the material by industry are therefore anticipated. Acknowledgements Dr. Y. Zhang and Mr. J. Steel carried out some of the erosion tests. This work was supported by National Natural Sciences Foundation of China, Monash University, Australia and Japan Society for the Promotion of Science. References 1. T. Ekström and M. Nygren, “Sialon Ceramics”, J. Am. Ceram. Soc., 75 (1992) 259-276. 2. A. Bonazza, L. Cunico, G. Dircetti, M. Dondi, G. Guarini and A. Ruffini, “Recycling of Steel Slag in Clay Brick Production, Key Engineering Materials”, 206-213 (2002) 835-838. 3. M. L. Ovecoglu, Microstructural Characterization And Physical Properties of a Slag-Based Glass-Ceramic Crystallized at 950 and 1100°C”, J. Eur. Ceram. Soc., 18 (1998) 161-168. 4. W.W. Chen, P.L. Wang, D.Y. Chen, B.L. Zhang, Y.B. Cheng and D.S. Yan, “Synthesis of (Ca, Mg)- α-Sialon from Slag by Self-propagating high-temperature synthesis”, J. Mater. Chem., 12 (2002) 1199-1202. 5. 5. Wei-Wu Chen, Joseph Steel, Yu Zhang, Jiu-Xin Jiang, Yi-Bing Cheng, Pei-Ling Wang and Dong-Sheng Yan, “Mechanical and Erosion-Resistance Properties of Slag α-Sialon Ceramics”, to be published in J. Eur. Ceram. Soc. 6. Y. Zhang, Y.B. Cheng and S. Lathabai, “Erosion of Alumina Ceramics by Air- and Water-Suspended Garnet Particles,” Wear, 241 (2000) 40-51. 7. Y. Zhang, Y.B. Cheng and S. Lathabai, ”Influence of Microstructure on the Erosive Wear Behavior of Ca α-Sialon Materials”, J. Euro. Ceram. Soc., 21 (2001) 2435-2445. Contact Details |