Introduction The preparation, characterization, and applications of organic/inorganic hybrid materials have become a fast expanding area of research in materials science. They combine the most important properties of two or more materials, forming a composite or bio-mineral system at the nanoscopic or molecular level [1-11].These hybrid sol-gel derived materials have been termed “CERAMERs” by Wilkes et al. and “ORMOSILs or ORMOCERs” by Schmidt et al. [1-3, 6, 11], and these terms have been used by many authors [2, 5-6, 12-29]. This type of material is easily accessible because of the commercial availability of a great number of precursors (alkoxides, organo(alkoxi)silanes and nanoparticles) [8, 17]. In addition to the simple metal or silicon alkoxides lead to inorganic oxide networks after the hydrolysis, organo(alkoxi)silanes can be used to incorporate polymerizable organic constituents such as methacryloxy, vinyl, allyl, alkyl, epoxy or other functional groups into the final products, because the Si-C bonds in these molecules are stable under the mild conditions of sol–gel processing [1, 4-5, 8]. Inorganic- organic materials have been roughly divided into three general classes: Class I corresponds to organic molecules, oligomers, prepolymers or even polymer, of low molecular weight which are simply embedded in an inorganic matrix. Both components exchange rather weak bonds, mainly through van der Waals, hydrogen, or ionic interactions. Class II corresponds to hybrid organic- inorganic compounds in which organic and inorganic components are bonded through stronger covalent or ionic- covalent chemical bonds [2, 5, 8, 19]. Class III consists of are biocomposites produced by natural biomineralization, where the inorganic phase is regularly and highly organized and grown in situ in the main organic polymeric under gentle conditions. The biomineralization process involves steps difficult to carry out completely in systems of artificial models; however, there have been various efforts to investigate them [7]. ORMOSILs with a wide variety of potential applications have been reported, such as coating with the following properties: antifogging [1,3]; abrasion resistant for polymers [1,6,8,11], dust-repellent [3], antisoiling [1]; hydrophilic [10], hydrophobic [11], antistatic films [8]; corrosion protective for metal surfaces [1,8,18], photochromic glass surfaces [3]; and color decorative for glasses and plastics [3,8]. They are used in bulk for electronic and electrical purposes [5,8,20]. In addition, those materials have applications such as nanoparticles, aerogels, bulk, nanocomposites and fibers [8,11]. Also there are many others types of hybrids: sandwich structures, foams, lattice structures and more [9]. Organic dyes have several advantages over pigments when they are used in colored thin films because of their higher intensity of light absorption and transparency, non scattering, high solubility [22-24], very wide availability, and great variety, there are more than 7000 types of them [25]. These characteristics of the organic dyes in many types of hybrid organic-inorganic matrixes can produce better distribution and homogeneity and intense color in the films, even in low thickness films. Organic dye in a hybrid organic-inorganic matrix has better characteristics than an organic matrix, because the dye colored hybrid films have enhanced mechanical properties like higher hardness [5,21] and stronger abrasion resistance [19]. With respect to inorganic sol-gel films, hybrid films also provide several advantages such as much better adhesion, transparency, flexibility, reduced surface roughness, and better refractive index matching [1-5]. The incorporation of the dye into ormosil matrix can be by means of weak bonds (class I) or by means of hards bond (class II); in the latter case the organic dye can be chemically modified to covalently attach them to the inorganic network through modification of silica gel glasses, obtained from tetraethylortosilicate (TEOS) with the organosiloxanos compounds and or surfactants [4,16,26]. For some years, the coloration by dye hybrid coatings has been applied to crystal glasses and plastics in industrial processes such as non-linear optics (NLO) [2,6-7], laser performance [13-14], holographic diffraction gratings [16], and decoration of transparent glass and crystal glassware [15,17,27]. In this paper we have taken advantage of the high solubility and hard bonds of organic dyes in a hybrid organic-inorganic polymethylmethacrylate-silica (PMMA-SiO2) matrix to obtain homogeneous, hard and high optical quality red, blue and green color films on glass substrates. The films were prepared by the sol-gel method with a composition: MMA (100%), SiO2 (100%) and 3-(trimethoxysilyl) propyl methacrylate (TMSPM) 50% mol with respect to TEOS. Experimental Hybrid polymethylmethacrylate-silica films with varying amounts of embedded dyes were prepared using tetraethyl ortosilicate (TEOS, 99%, Aldrich) as the silica source, methyl methacrylate (MMA, 99%, Aldrich), 3-(trimethoxysilyl)propylmethacrylate (TMSPM, 98% Aldrich), ethanol (EtOH), as solvent, distilled deionized water, HCl, benzoyl peroxide (BPO, Aldrich) as catalyst and three organic dyes (red, green and blue), supplied by Mardupol Chemicals Company. The precursor solution was prepared as follows: TMSPM was added to a pre-polymerized TEOS solution of ethanol and then the substiochiometric amount of water (diluted HCl, pH 2) was added drop-wise while stirring the solution was then stirred at room temperature for 1 h. For the organic component, BPO was added to the MMA system and stirred at room temperature for 1 h. After that, both solutions were mixed. The precursor solution to deposit the hybrid coatings was prepared using a 1:0.5:1 molar ratio of reactants for TEOS:TMSPM:MMA respectively. On the other hand, organic dye dispersions were prepared in ethanol from the commercial powder red, green and blue dyes. To obtain different color intensities, different amounts of the dye dispersion were added to the hybrid precursor solution; 0.17% wt for blue; 0.34% wt for red; and 0.5 mass%, for green. After that, the resulting solution in each case was stirred at room temperature for 24 h. The hybrid films were deposited by the dip-coating method on pre-cleaned microscope glass slide substrates. Four types of coatings were deposited and studied: uncolored (UC), blue (BC), green (GC) and red (RC) transparent hybrid coatings. All the obtained hybrid films were studied by FTIR spectroscopy. These measurements were performed with a Nicolet Avantar 360 FTIR spectrometer. Images of the films surfaces were obtained by using an atomic force microscope Scanning Microprobe SPC-400 from Park Instruments. The optical reflectance and transmittance measurements at normal incidence were carried out in the UV-visible spectral range from 240 to 840 nm, using a thin film metrology system, FilmTekTM 3000, Scientific Computing International (SCI), Inc. The hardness of the coatings was estimated according to the pencil hardness test referring to ASTM Standard D 3363- 92 [28]. Results and Discussion The infrared spectra of the UC, BC, RC and GC hybrid films are shown in Figure 1. The four spectra were normalized to the corresponding maximum value of the absorption and they are arbitrarily displaced along the absorption axis. At the bottom, the spectrum of the colorless 0C hybrid is very similar to those of hybrid films reported in the literatures [29-31]. It displays intense absorption peaks at about 954, 1080, 1170 cm-1, and a broad band between 3100 and 3600 cm-1, associated with the absorption of the Si-OH group, transversal optic (TO) Si-O-Si asymmetric stretching, longitudinal optic (LO) Si-O-Si asymmetric stretching and hydroxyl groups, respectively. In addition, a weak signal is observed at about 800 cm-1, which is related to the absorption by Si-O-Si symmetric stretching. The appearance of the first peak indicates an incomplete condensation of the Si-OH bond in the hybrid matrix, and the peaks due to the Si-O-Si asymmetric and symmetric stretching are evidence of the formation of the silica network by the sol-gel process. The spectra of the BC, GC and RC films also display absorption peaks due to the silica structure and those due to the silanol and hydroxyl groups. In these spectra there are additional similar weak absorption signals produced by the organic dye in the three colors. It is also very evident that the ratio of the intensity of the peak at 1080 cm-1 to that of the peak at 1170 cm-1 is smaller in the spectra of the colored films. The increase of the second peak relative to the first one attributed to an increase in the porosity of the SiO2 matrix [32]. It has been reported that the organic-inorganic interfacial interaction can be initiated through the formation of hydrogen bonds [29,30]. In our case, the residual silanol groups in the silica structure are capable of forming a hydrogen bond and their presence is indicated in the hybrid films by the absorption peak at 954 cm-1 and the broad absorption band due to the hydroxyl groups in the FTIR spectra of four hybrid films. Thus, the infrared measurements are compatible with the formation of hybrid films with a homogeneous matrix constituted by both organic and inorganic components with embedded organic dye molecules. | 
| | Figure 1. FTIR spectra of the uncolored and colored hybrid films. | In Figures 2 a), 2 b) and 2 c) are shown 3-D AFM images taken from a 5 x 5 mm2 area in the surface of the UC, BC, and GC hybrid films, respectively. The morphology of the surface is very similar for the three hybrid films, showing very smooth and flat surfaces with very little roughness. The values of the rms average roughness measured for the three films was 0..47, 0.65 and 0.45 nm, respectively. There is no evidence of dye aggregation in the surface of the colored hybrid films, even in the two hybrid film dye concentration, since their granular structure is very similar to that of the uncolored film. These results suggest that the organic dye is very well dispersed in the entire hybrid matrix. The surface hardness of the hybrids films was measured by a pencil test according to the ASTM Standard D 3363- 92 [40]. | 
(a) 
(b) 
(c) | | Figure 2. AFM images showing the morphology of the colored SiO2-PMMA hybrid coatings with molar ratio formulation of: 1:0.5:1.0 TEOS:TMSPM:MMA respectively, with different concentrations and types of color: a) 0 mass% of color, incolor b) 0.17% Wt of Blue color, and c) 0.83% Wt of Green color. | The application of this test showed that the hardness of all the hybrid films is at least 9H. This result shows that the inorganic component in the hybrid films has reinforced the organic component, producing harder films with more abrasion resistance as compared with pure PMMA films. Figure 3 shows the optical spectra of transmittance (T) and reflectance (R) for the BC, RC and GC film in a range covering the visible spectra. The continuous lines are the experimental data and the dashed lines are the best-fit spectra calculated according to a model described below. Both R and T spectra show an absorption band in the range of about 489 to 713 for BC, 420 to 600 nm for RC, and 397 to 497 and 540 to 687 for GC; out of this range the T spectrum shows values of about 90 % for the three hybrid coatings. The interference oscillations which are present indicate the good optical quality of the three films. The optical transparency of our hybrid films is another indication of their homogeneous composition of both organic and inorganic phases. This is because when both phases separate at the macroscopic level in the hybrid matrix, the inorganic oxide phase scatters the light, producing opaque hybrid films as a result. In order to analyze the experimental spectra of Figure 3, an air/coating/substrate system was considered. For that, the optical response of the hybrid films was represented by a generalized form of the Lorentz harmonic oscillator expression, patented by SCI, which is included in the software of the optical measurement system [33]. In this case, the parameters of three oscillators were used plus two global parameters, the high energy dielectric constant (ε∞) and the damping coefficient (α), considered in the SCI-model [33]. It can be clearly observed in this graph that this model appropriately describes the experimental spectra and that similar results were obtained for all the samples analyzed. The optical constants, refractive index (n) and extinction coefficient (k), obtained for the three hybrid films, are shown in Figure 4. Two of the three oscillators representing the absorption due to the dye are centered at 2.05 and 1.94 eV in BC, which correspond to 605 and 640 nm, respectively, 2.25 and 2.37 eV (551 and 523 nm) in RC, and 2 and 1.96 eV (620-632 nm) in VC . The third high energy oscillator represents the optical response of the matrix. As can be seen in Figure 4, the n spectrum shows a mild wavelength dispersion with and has values of about, 1.36, 1.41 and 1.42 for BC, VC and RC, respectively. The thicknesses of the films are of 1350 nm, 1808 nm and 1492 nm, for BC, RC and VC films, respectively. 
Figure 3. Transmittance and reflectance spectra of the three colored hybrid film. The color produced by introducing the dye into the hybrid transparent matrix is an important issue to be quantified. Therefore, the color properties were analyzed using the CIE Colorimetric System, CIE 1931 2-degree Standard Observer [34]. The purpose of this analysis is to give color specifications for observers with normal vision in terms of tristimulus values or chromaticity coordinates. For that, considering the distribution functions 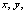 and and 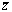 , that represent the red, green, and blue response of the standard eye, respectively, the CIE tristimulus values (XCIE, YCIE, ZCIE), corresponding to a certain color stimulus ψ(λ) are obtained from , that represent the red, green, and blue response of the standard eye, respectively, the CIE tristimulus values (XCIE, YCIE, ZCIE), corresponding to a certain color stimulus ψ(λ) are obtained from 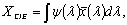 (1) (1)
and analogous expressions for YCIE and ZCIE. The color stimuli are ψ(λ)=S(λ)T(λ) where S(λ) is the spectral irradiance function of the illuminant, for which for the present case the average daylight or illuminant C was used. Because the color of the hybrid films is dominated by the optical absorption produced by the dye, the transmittance spectra T(λ) is used to calculate the color stimuli. Chromaticity coordinates are obtained from 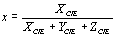 (2) (2)
and correspondingly for y and z. Thus, the x and y coordinates obtained for the three dye coatings are given in Table 1, and are plotted in the chromaticity diagram of Figure 5 (a). In this figure, the coordinates for the BC, RC and VC films are indicated by the arrows. The latter is shown in Figure 5 (b), which is a magnification of the x-y region of interest; the coordinates x=0.3101 and y=0.3163 for the illuminant C are also shown. Table 1. CIE chromaticity coordinates and CIELAB parameters for the four blue, red and green hybrid films | | | BC | -10.518 | -17.118 | 86.113 | 0.264 | 0.290 | | RC | 24.383 | -6.343 | 86.784 | 0.333 | 0.290 | | VC | -15.384 | -5.176 | 91.173 | 0.280 | 0.317 | | 
| | Figure 4. Optical constants n and k for the three colored hybrid films. | | 
| | Figure 5: (a) Chromaticity coordinates for the three colored hybrid films. (b) Magnification of (a). (c) CIELAB coordinates for the colored films in the a*-b* plane. | Because the CIE chromaticity diagram does have a few drawbacks, namely that, brightness is difficult to include and that there is a discrepancy between perceived color differences and the actual spacing of color in the system, another common approach used for human color perception is the CIELAB system [35], which consists of three color coordinates: brightness L*, the red-green value a*, and the yellow-blue value b*. The corresponding CIELAB coordinates for the hybrid films were calculated from the tristimulus values, and are listed in Table 1. Conclusions In this paper we have reported a sol–gel method for the deposition of red, green and blue PMMA-SiO2 hybrid films on glass substrates. We found that the colored hybrid films are composed of a homogeneous organic-inorganic matrix with the embedded organic dye molecules very well dispersed on it. The microscopy measurements showed that the surface of the films is very flat and smooth with very little roughness and reinforced hardness in comparison with the pure PMMA phase. The transparent colored hybrid films have a very good optical quality with color intensity depending on the amount of organic dye in the films. The color coordinates of the hybrid films were determined for all three colored films studied in the CIE and CIELAB systems. References 1. R. Kasemann and Schmidt, “Coatings for mechanical and chemical protection based on organic-inorganic sol-gel nanocomposites”, New J. Chem., 18 (1994) 1117-1123. 2. C. Sanchez and F. Ribot, “Design of hybrid organic-inorganic materials synthesized via sol-gel chemistry”, New J. Chem., 18 (1994) 1007-1047. 3. H. Schmidt, “Multifunctional inorganic–organic composite sol–gel coating for glass surfaces”, J. Non-Crystalline Solids, 178 (1994) 302-312. 4. U. Schubert, N. Hüsing and A. Lorenz, “Hybrid Inorganic-Organic Materials by Sol-Gel Processing of Organofunctional Metal Alkoxides”, Chem. Mater., 7 (1995) 2010-2027. 5. P. Judeinstein and C. Sanchez, “Hybrid organic-inorganic materials: a land of multidiciplinarity”, J. Mater. Chem., 6 [4] (1996) 511-525. 6. J. Wen and G. L. Wilkes, “Organic/Inorganic Hybrid Network Materials by the Sol–Gel Approach”, Chem. Mater., 8 (1996) 1667-1681. 7. S. Yano, K. Iwata and K. Kurita, “Physical properties and structure of organic–inorganic hybrid materials produced by sol–gel process”, Materials Science and Engineering, C 6 (1998) 75-90. 8. G. Schottner, “Hybrid Sol–Gel-Derived Polymers: Applications of Multifunctional Materials”, Chem. Mater., 13 (2001) 3422-3435. 9. M. F. Ashby and Y. J. M. Bréchet, “Designing hybrid materials”, Acta Materalia, 51 (2003) 5801-5821. 10. K.-Ch. Song, J.-K. Park, H.-U. Kang and S.-H. Kim, “Synthesis of Hydrophilic Coating Solution for Polymer Substrate Using Glycidoxypropyltrimethoxysilane”, J. Sol-Gel Science and Technology, 27 (2003) 53-59. 11. J. D. Mackenzie, “Sol-Gel Research—Achievements since 1981 and Prospects for the Future”, J. Sol-Gel Science and Technology, 26 (2003) 23-27. 12. C. J. Brinker and G. W. Scherer, “Sol-Gel Science: The Physics and Chemistry of Sol–Gel Science: The Physics and Chemistry of Sol–Gel Processing”, Academic Press, New York (1990) pp. 115, 866. 13. M. D. Rahn and T. A. King, “Comparison of laser performance of dye molecules in sol-gel, polycom, ormosil, and poly(methyl methacrylate) host media”, Applied Optics, 34-36 (1995) 8260-8271. 14. M. Casalboni, F. De Matteis, P. Prosposito and R. Pizzoferrato “Optical investigation of infrared dyes in hybrid thin films”, Applied Physics Letters, 75-15 (1999) 2172-2174. 15. J. Kron, G. Schottner, K. -J. Deichmann, “Glass design via hybrid sol-gel materials”, Thin Solids Films, 392 (2001) 236-242. 16. I. G. Marino, D. Bersani and P. P. Lottici, “Holographic gratings in DRI-doped sol-gel silica and ORMOSILs thin films”, Optical Materials, 15 (2001) 279-284. 17. J. Zaccaro, N. Sanz, E. Botzung, Appert, P. L. Baldeck and A. Ibanez, “Organic nanocrystals grown in sol-gel matrices: a new type of hybrid material for optics”, C. R. Physique, 3 (2002) 463-478. 18. T.P. Chow, C. Chandrasekaran and G.Z. Cao, “Sol-Gel-Derived Hybrid Coating for Corrosion Protection”, J. Sol-Gel Science and Technology, 26 (2003) 321-327. 19. J. D. Mackenzie and Eric Bescher, “Some Factors Governing the Coating of Organic Polymers by Sol-Gel Derived Hybrid Materials”, J. Sol-Gel Science and Technology, 27 (2003) 7-14. 20. F. De Matteis, P. Prosposito, M. Casalboni, M. L. Grilli, E. Di Bartolomeo and E. Traversa, “Electrical Properties of Sol-Gel Processed irbid Films”, J. Sol-Gel Science and Technology, 26 (2003) 1081-1084. 21. O. A. Shilova, S. V. Hashkovsky and E. V. Tarasyuk, “Organic-Inorganic Coatings Based on Sol-Gel Technology”, J. Sol-Gel Science and Technology, 26 (2003)1131-1135. 22. D. Malacara, “Color Vision and Colorimetry, Theory and Applications”, SPIE PRESS, Washington (2002) pp. 113-114. 23. J. T. Lutz Jr. and R. F. Grossman, “Polymer Modifiers and Additives”, Marcel Decker Inc., New York, (1988) pp. 35. 24. W. Herbst and K. Hunger, “Industrial Organic Pigment, Production, Properties, Applications”, VCH, Germany, (1997) pp. 1. 25. R. J. D. Tilley, “Colour and Optical Properties of Materials, An Exploration of the Relationship between Light, the Optical properties of Materials and Colour”, John Wiley & Sons LTD, London, (2000) pp. 194. 26. Łączka, K. Cholewa-Kowalska and M. Kogut, “Organic-inorganic hybrid glasses of selective optical transmission”, J. of Non-Crystalline Solids, 287 (2001) 10-14. 27. G. Schottner, J. Kron and A. Deichmann, “Industrial Application of Hybrid Sol-Gel for the Decoration of Crystal Glassware”, J. Sol-Gel Science and Technology, 13 (1998) 183-187. 28. The American Society for Testing and Materials D 3363- 92 Test Method for Film Hardness by Pencil Test. 29. Z.H. Huang and K.Y. Qiu, “The effects of interactions on the properties of acrylic polymers/silica hybrid materials prepared by the in situ sol-gel process”, Polymer, 38-3 (1997) 521-526. 30. C.K. Chan, S.L. Peng, I.M. Chu and S.C. Ni, “Effects of heat treatment on the properties of poly(methyl methacrylate)/silica hybrid materials prepared by sol-gel process”, Polymer, 42 (2001) 4189-4196. 31. J. Gallardo, A. Duran, D. Di Martino and R. M. Almeida, “Structure of inorganic and Irbid SiO2 sol-gel coatings studied by variable incidence infrared spectroscopy”, J. Non-Crystalline Solids, 298 (2002) 219-225. 32. A. Fidalgo and L.M. Ilharco, “Thickness, Morphology and Structure of Sol-Gel Hybrid Films: I-The Role of Precursor Solution’s Aging”, J. Sol-Gel Science and Technology, 26 (2003) 363-367. 33. E. Zawaideh, “Nondestructive Optical Techniques for Simultaneously Measuring Optical Constants and Thicknesses of Single and Multilayer Films”, U. S. Patent No. 5889592 (30 March1999). 34. C.R. Bamford, “Colour Generation and Control in Glass (Glass science and technology)”, Elsevier, Amsterdam (1977) pp.16. 35. D. Malacara, “Color Vision and Colorimetry, Theory and Applications”, SPIE - The International Society for Optical Engineering PRESS, Washington (2002) pp.90-101. Contact Details |