| The component elements in steel can be categorised in terms of their crystal structures. At least a basic knowledge of the practical implications of these crystal arrangements is essential to understand the performance of steel in service. The structures are dependent upon the concentrations of each element, the fashion in which the steel is cooled from furnace temperatures, and the amount of cold work performed on the steel. Crystal Structures Ferrite Ferrite (α), is the crystal arrangement for pure iron. This form exists as part of the structure in most steels and can usefully absorb carbides of iron and other metals by diffusion in the solid state. Ferrite takes a body centred cubic (bcc) form and is soft and ductile. Austenite Austenite (γ), is a solid solution, that is, the component elements are arranged as if in solution (it also exists as an allotrope of pure iron). All steel exists in this form at sufficiently high temperatures (see figure 1). Some alloy steels stabilise this singular phase and it is present even at room temperatures. The crystal arrangement is face centred cubic (fcc) and, like ferrite, it is soft and ductile. Cementite Cementite is iron carbide (Fe3C), When carbon atoms can no longer be accommodated in solution in ferrite and austenite (due to an increase in carbon content or reduction in temperature), cementite forms, as it can accommodate more carbon in its crystal structure. Like other carbides, it is hard and brittle. Pearlite
Pearlite is a phase mixture consisting of alternating platelets of ferrite and cementite (α + Fe3C), which grows by conversion from austenite. A steel containing 0.77 wt% carbon can consist solely of pearlite if cooled sufficiently slowly from austenite (see figure 1). Under the microscope it can have an iridescent mother of pearl appearance, hence the name. Martensite Martensite is commonly found in steel that has been rapidly cooled ('quenched') from austenite. It is a particularly hard, brittle arrangement. Essentially it forms because any carbon in solid solution in the austenitic phase at high temperatures does not have enough time to be incorporated into cementite when cooled rapidly. The austenite crystals undergo a transformation involving the shearing of atom planes over each other. Martensite does not appear on the phase diagram (figure 1), as it is not an equilibrium phase. The strain energy involved in the martensitic reaction is enormous and a large undercooling is necessary. In low and medium carbon alloys, the martensite tends to form in lath shaped crystals that are generally too fine to resolve in the light microscope. In high carbon steels, plate martensite forms. For certain steels, the rapid cooling necessary to produce a martensitic structure (e.g. water or brine baths) introduces large surface tensile stresses and may cause quench cracking. However, when medium carbon steels are alloyed with elements such as nickel, chromium and molybdenum, the development of equilibrium phases is suppressed and martensite can be formed with less drastic cooling, such as oil quenching. Bainite If the steel is cooled such that the formation of pearlite by the short range diffusion of iron atoms is not possible, bainite can be produced. The bainite that forms at temperatures just below those at which pearlite forms is termed upper bainite. At lower temperatures, lower bainite forms. Both lower and upper bainite consist of aggregates of platelets or laths of ferrite, separated by regions of residual phases consisting of untransformed austenite or of phases such as martensite or cementite | 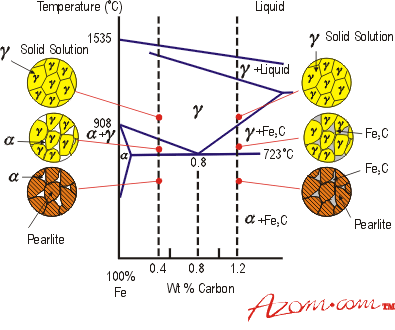
| | Figure 1. Part of the equilibrium diagram for the Fe-C system | |