Dec 5 2014
A research team at the University of Illinois at Urbana-Champaign has developed a low-cost hydrolyzable polymer by reversing the properties of polyurea, an important bonding material. This breakthrough offers an alternative solution to packaging, tissue engineering, and drug delivery.
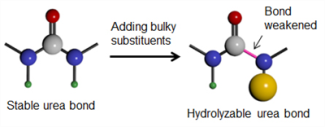 Hindered urea bond-containing polymeric materials (PHUs) are cheap polymers that can be designed to degrade over a specified time period. Image: Catherine Yao.
Hindered urea bond-containing polymeric materials (PHUs) are cheap polymers that can be designed to degrade over a specified time period. Image: Catherine Yao.
Hydrolyzable polymers have been used in a wide range of biomedical applications, such as transient medical implants/devices and surgical sutures. Polyurea materials are commonly used in painting, coating, and adhesives. It is extremely stable, thanks to the highly inert urea-bond, making it ideal for long-lasting applications.
The research team leveraged inventive chemistry to create a class of poly(hindered urea)s (PHUs), also known as hindered urea bond-containing polymeric materials. These polymers are not only low cost, but can also be made to degrade over a given time period, thus making them extremely useful in agricultural and biomedical applications.
PHUs can be hydrolyzed within a matter of days, unlike standard polyurea that are extremely stable against hydrolysis. Given that ‘hindrance’ is the major cause of bond destabilization, PHUs’ hydrolysis kinetics can be adjusted to suit a particular application. PHUs can possibly serve as eco-friendly and sustainable materials.
The study shows how PHUs can be leveraged to design polymeric materials that are water degradable and can be easily produced by combining multifunctional isocyanates and amines to extend the group of hydrolyzable polymers.
Hydrolyzable polymers have also been applied in the design of controlled release systems in agriculture and food industries and used as degradable, environmentally friendly plastics and packaging materials. These applications usually require short functioning time, complete degradation and clearance of materials after their use.
Jianjun Cheng
Associate Professor of Materials Science and Engineering
University of Illinois
In addition, the new PHUs offer significant benefits when compared to other hydrolyzable polymers. They can be developed with low-cost chemical precursors in ambient conditions through clean chemistry without any by-products or the need for a catalyst.
This allows end-users to exploit the copolymer recipe for certain applications without the necessity for complex synthesis apparatus.