Apr 1 2016
Scientists at the U.S. Department of Energy's Argonne National Laboratory have demonstrated that the design and placement of a tiny measurement device called a reference electrode enhances the quantity and quality of information that can be extracted from lithium-ion battery cells during cycling.
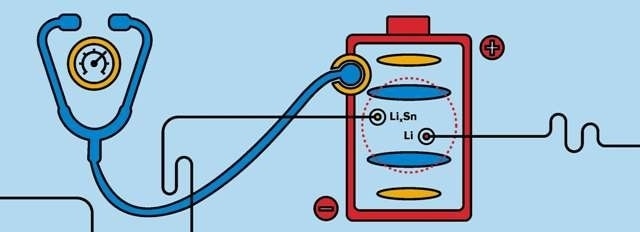 Reference electrodes provide insights into a battery cell's health. The image above depicts two reference electrodes within a battery cell. This configuration that allows researchers to evaluate a battery's anode and cathode separately at all stages of cycling and aging. (credit: Sana Sandler/Argonne National Laboratory)
Reference electrodes provide insights into a battery cell's health. The image above depicts two reference electrodes within a battery cell. This configuration that allows researchers to evaluate a battery's anode and cathode separately at all stages of cycling and aging. (credit: Sana Sandler/Argonne National Laboratory)
Reference electrodes (REs) are used to measure the voltages of individual electrodes that make up the battery cell. "Such information is critical, especially when developing batteries for larger scale applications, such as electric vehicles, that have far greater energy density and longevity requirements than typical batteries in cell phones and laptop computers," said Argonne battery researcher Daniel Abraham, co-author of a newly published study in the Journal of the Electrochemical Society. "This kind of detailed information provides insight into a battery cell's health; it's the type of information that researchers need to evaluate battery materials at all stages of their development."
Argonne battery researchers have been at the forefront of using reference electrodes to evaluate the performance of lithium-ion cells, Abraham said. Their studies have provided crucial insights into cell aging phenomena, including the effects of test temperatures and cycling voltages. Mitigating the root causes of aging can increase cell longevity and improve the commercial viability for applications that require long-term battery durability.
Until recently, Argonne battery researchers would use only one reference electrode, based on a lithium-tin (Li-Sn) alloy, to collect information. However, Abraham's team found that by sandwiching a Li-Sn reference electrode between the positive and negative electrodes, while simultaneously positioning a pure lithium metal reference electrode next to the stack, they could obtain insights into electrode state-of-charge shifts, active material use, active material loss and impedance changes.
In testing the new reference electrode configuration, researchers used a cell containing a lithiated oxide cathode (NCM-523), an Argonne-developed silicon-graphite anode (Si-Gr) and various electrolytes, including ones with fluoroethylene carbonate (FEC) or vinylene carbonate (VC) additives. Both NCM-523 and Si-Gr are materials of interest for high-energy-density lithium-ion batteries being developed to extend the driving range of vehicles.
"Silicon-containing electrodes could double the energy stored in lithium-ion cells," said Abraham. But because silicon-containing cells degrade more quickly, the Argonne team wanted to know the impact of the FEC and VC addition to the cell electrolyte. "Our new reference electrode configuration confirms the beneficial impact of these additives, not only in reducing capacity loss but also in mitigating the impedance rise displayed by cells without these additives," he added.
This research was funded by the U .S. Department of Energy's Vehicle Technologies Office.