Nov 2 2016
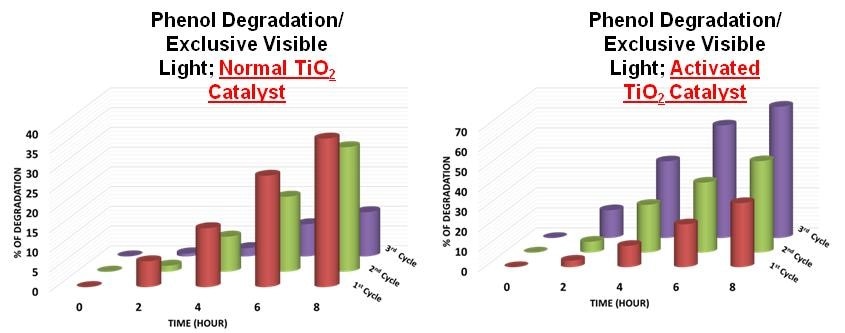 This is a stack plot showing the percent of degradation of Phenol vs Time for (A) TiO2 and (B) TiO2-1W for all the three cycles Credit: Sorathiya et al
This is a stack plot showing the percent of degradation of Phenol vs Time for (A) TiO2 and (B) TiO2-1W for all the three cycles Credit: Sorathiya et al
A catalyst is used in chemistry, to achieve the efficiency of any chemical reaction. This catalyst helps to increase the rate of reaction without causing any harmful effects.
Specifically, a photocatalyst is activated under light and can lead to improvements in rates of a wide range of redox based reactions. Recyclability is one aspect of photocatalysis that is not often recognized.
A catalyst is only considered viable when it is able to repeatedly withstand the reaction conditions and the rates of reaction, when re-using the catalysts, must not undergo dramatic changes (or any changes at all). However, in reality many catalysts ultimately degrade.
Deepa Khushalani's group has presented a seminal observation in which an increase in rate of reaction upon recycling was detected. When compared with the first cycle, the observed increase was 1.7 times higher in the second cycle and 3.1 times higher in the third cycle.
The group demonstrated that photocatalysts are capable of undergoing structural evolution and, in specific conditions, develop into a more efficient additive for consequent reusability. This feature seems to be unique as the understanding, previous to this research, was that catalysts continue to be unchanged during the reactions.