Apr 25 2019
Compared to conventional lithium-ion batteries, batteries containing metallic lithium anodes deliver better efficiency due to their higher capacity. However, safety issues and a short service life hinder its path. To properly examine the causes of glitches and premature failure of such batteries, scientists have formulated a method that visualizes the distribution of active lithium on the anode and distinguishes between dendrites and “dead” lithium. As mentioned in the journal Angewandte Chemie, the method utilizes a fluorescent dye.
 © Wiley-VCH
© Wiley-VCH
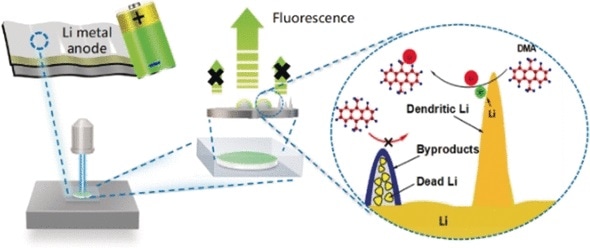
As a lithium anode battery discharges, the anode emits electrons to the circuit and positively charged lithium ions to the electrolyte. When the battery is recharged, the process is reversed, placing lithium back onto the anode. Regrettably, deposition is not even and can result in the development of branched structures called dendrites, which can grow so large that they result in a short circuit. Furthermore, their higher surface area increases unfavorable side reactions between lithium and the constituents of the electrolyte, which neutralizes the lithium. In the end, a few dendrites consist totally of this “dead” lithium. Although both dead lithium and dendrites obstruct the power of the battery, they each have a totally different effect on the anode. As the morphology is identical in both cases, it has not formerly been feasible to distinguish between them with conventional microscopy methods.
To better comprehend the unfavorable processes that take place at lithium anodes, scientists working with Shougang Chen, Shanmu Dong, and Guanglei Cui at the Chinese Academy of Sciences and the Ocean University of China in Qingdao (China), have currently formulated a novel method that enables them to examine the distribution of active lithium species on the surface of the anode and to distinguish between lithium dendrites and side products.
The surfaces of used lithium anodes have a coating of a fluorescent dye called 9,10-dimethylanthracene (DMA). Lithium responds to DMA, quenching its fluorescence. Areas with active lithium, thus, appear dark, while areas that have inactive lithium species continue to fluoresce. The anode’s morphology is not impacted.
For lithium metal batteries to be used in a safe manner, it is very crucial to identify the reasons behind the potentially hazardous malfunctions. With this new technique, it is viable to spot dendrites that have led to the faulty operation of a lithium battery. In the creation of new batteries, this method also helps the search for enhanced electrolytes and offers predictions concerning the uneven deposition of lithium. Locating the sites where lithium dendrites develop preferentially may help to enhance the structure of new batteries.