It has been reported that hemorrhage accounts for around half of battlefield deaths, with almost 86% of deaths in combat occurring within 30 minutes of wounding.
While it is comparatively easier to treat wounds that are external and immediately visible, internal hemorrhages or abdominal wounds represent a distinct challenge because they are difficult to locate without specific medical equipment.
If immediate responders are to successfully treat trauma wounds and damage to the liver, stomach, colon, spleen, venous, arterial, or other hard-to-reach systems and areas, a hemostatic treatment must be employed.
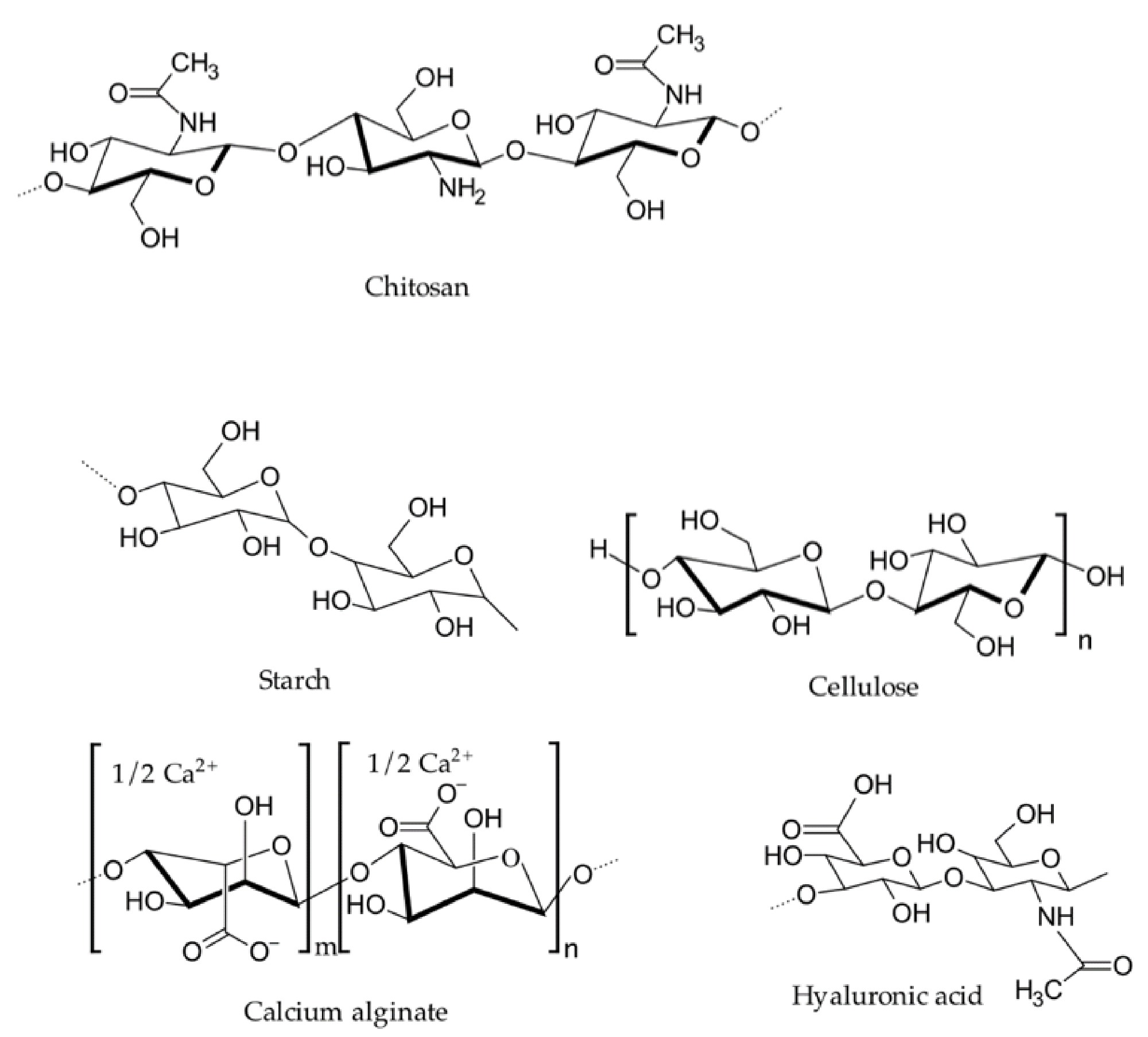
Representative chemical structures of naturally occurring hemostatic polymers. Image Credit: Ghimire, S et al., Pharmaceutics
Ideally, these treatments would be FDA-approved, able to stop bleeding within the first 1 to 2 minutes of wounding, and applicable in under 30 seconds; all without the need to apply manual pressure to the wound area.
While blood transfusions remain the most direct treatment for a severe loss of blood, these are not practical in the field due to unsterile conditions, environmental factors, and a lack of specialist equipment.
Wound dressings offer an alternate means of preventing blood loss and encouraging healing, but these too run the risk of infection or even sepsis unless an appropriately sterile environment can be ensured.
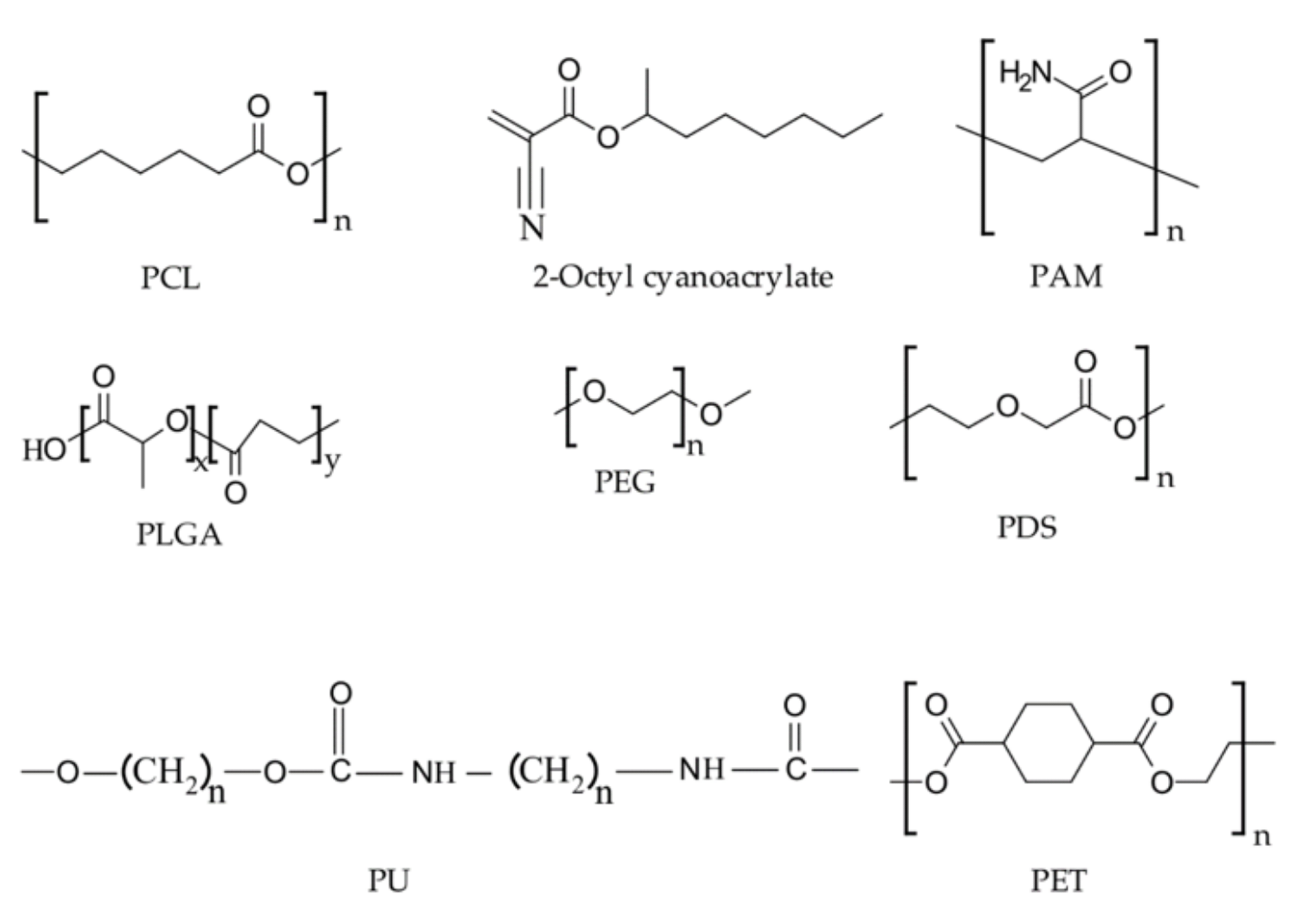
Representative chemical structures of synthetic hemostatic polymers. Image Credit: Ghimire, S et al., Pharmaceutics
To help overcome the limitations in the field application of these methods, considerable research has been undertaken into the design and fabrication of hemostatic wound-healing materials – materials able to halt blood flow from a wound site by mimicking the body’s natural healing process.
Polymers – both natural and synthetic - have attracted attention as potentially viable options for hemostatic materials, offering a number of advantages over biologically-derived hemostatic materials including significantly reduced risk of disease transmission and improved biocompatibility.
The paper’s authors aimed to collate and review current work into the use of polymers in the development of hemostatic materials, highlighting the past two decades’ advancements and providing a number of examples of polymers’ successful application in this field.
The authors begin by outlining the scope of good hemostatic material, highlighting that this should be high-strength, biodegradable, and have excellent tissue compatibility. It should also offer useful wound dressing properties.
With this in mind, they evaluated and investigated research into a number of natural polymers – including chitosan, collagen, gelatin, alginate, and oxidized cellulose – and a range of synthetic polymers – including polyesters, polycyanoacrylates, polyalkyleneoxides, polyethylene glycol, and polyurethane.
For each of these materials, they summarized recent research into their application in hemostatic materials, example studies, and the scope for further research.
Overall, the study found that the induction of sufficiently rapid hemostasis at a wound site remained a challenge for polymer-based hemostatic materials, particularly where there was a significant hemorrhage with high rates of bleeding. There were also found to be limited in terms of polymers’ adhesion strength, mechanical durability, elasticity, and other critical factors.
It was identified that there was significant work to be done in terms of developing hemostatic polymer materials that were efficient, mechanically strong, resilient, biocompatible, and able to achieve rapid hemostasis.
A number of polymer-based hemostatic materials are currently in development, including products packaged in a granular form able to be applied directly to wounds and products delivered as adhesive foam. Each method of application has specific advantages and disadvantages, particularly when deployed outside of a traditional medical setting.
The most prominent area of development for hemostatic materials lies in the use of hemostatic agents for prehospital hemorrhage control. Flowable hemostats are constantly being developed and improved in terms of their ability to work fast and improve visibility, with advanced flowable hemostats able to control bleeding from hard-to-reach sites in the shortest possible time.
Work in this area continues to be wide-ranging and rapidly advancing, and the authors conclude their comprehensive review by suggesting some future areas of work to help develop this field – specifically in terms of developing materials that not only have excellent hemostatic properties but that are also able to meet the strict regulatory requirements which must be met prior to use in a medical or other setting.
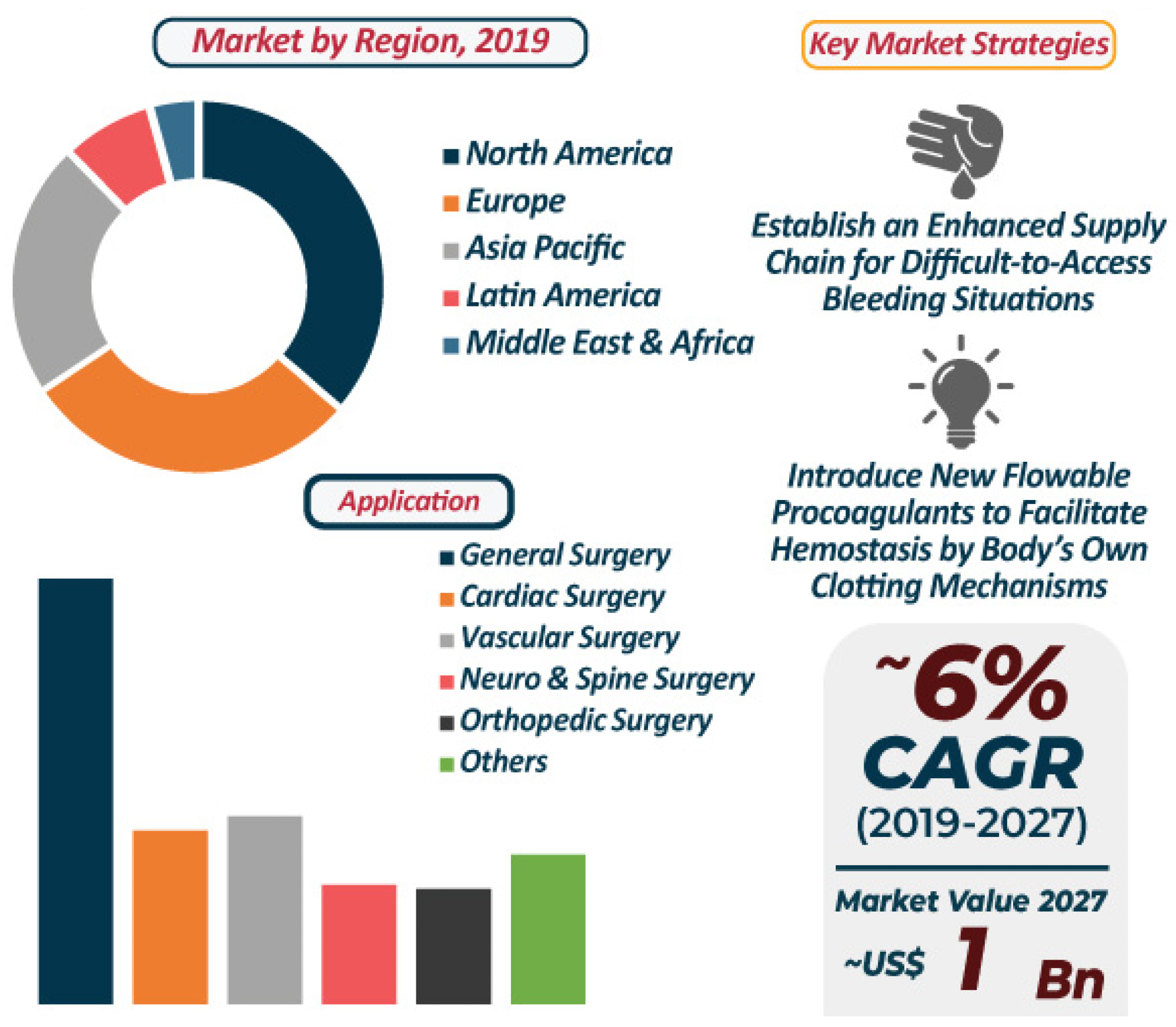
Flowable Hemostats market: 2019–2027. Image Credit: Ghimire, S et al., Pharmaceutics
A hemostatic material must provide the body with a means of quickly slowing or stopping bleeding from external and internal wounds. Current approaches face a number of issues including the risk of antibody response, the transmission of blood-borne pathogens, and thromboembolic complications.
Hemostatic materials must be able to avoid these risks while being able to function for extended periods before needing to be replaced – a particular issue with complex internal wounds which make it challenging to reapply materials.
The authors also highlight that the majority of the studies they investigated were done in controlled and lab-based settings, pointing out the need for analysis in as close to ‘real world’ scenarios as possible to truly develop and optimize viable and widely applicable hemostatic materials.
References
Ghimire, Suvash, Pritha Sarkar, Kasey Rigby, Aditya Maan, Santanu Mukherjee, Kaitlyn E. Crawford, and Kausik Mukhopadhyay. 2021. "Polymeric Materials for Hemostatic Wound Healing" Pharmaceutics 13, no. 12: 2127. https://www.mdpi.com/1999-4923/13/12/2127
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.