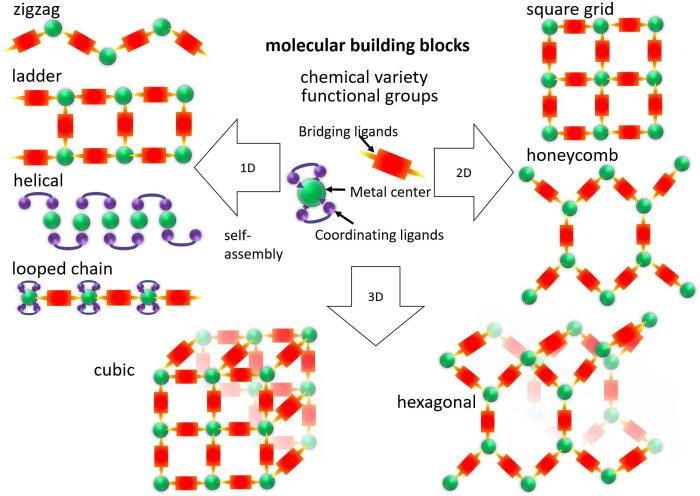
Building blocks of CPs and their assembly into secondary structures. Image Credit: G. H. Morritt et al., Chem. Phys. Rev.
It has been historically understood that polymers do not conduct electricity – a view primarily informed by research into and applications of plastics, arguably the most common form of polymer. The fact that plastics do not conduct electricity is often central to their use, for example, in the casings of electrical appliances or as casings for conductive cabling.
More recent advances have challenged these ingrained beliefs and understandings, however. The paper’s authors highlight the work of A. Heeger, A. MacDiarmid, and H. Shirakawa, who were awarded the Nobel Prize in Chemistry in 2000 for their work on conductive plastics.
This work represented the foundation of research into conductive polymers and the related field of molecular electronics based on the use of organic materials in electronics applications.
While the use of organic polymers has proved highly beneficial to the development of printable electronics, it was noted by the paper’s authors that there remained scope for improvement, for example, by introducing metal ions between organic molecular units to enhance their conductivity.
Coordination polymers represent another means of improving the performance of these polymers. Coordination polymers are created from metal centers connected by organic or inorganic bridging ligands, allowing the conduction of charge along the polymer chains in most instances.
Ongoing research into the application of coordination polymers in molecular electronic devices has led to the development of conductive coordination polymers that can employ organic linkers that function as hybrid organic/inorganic materials.
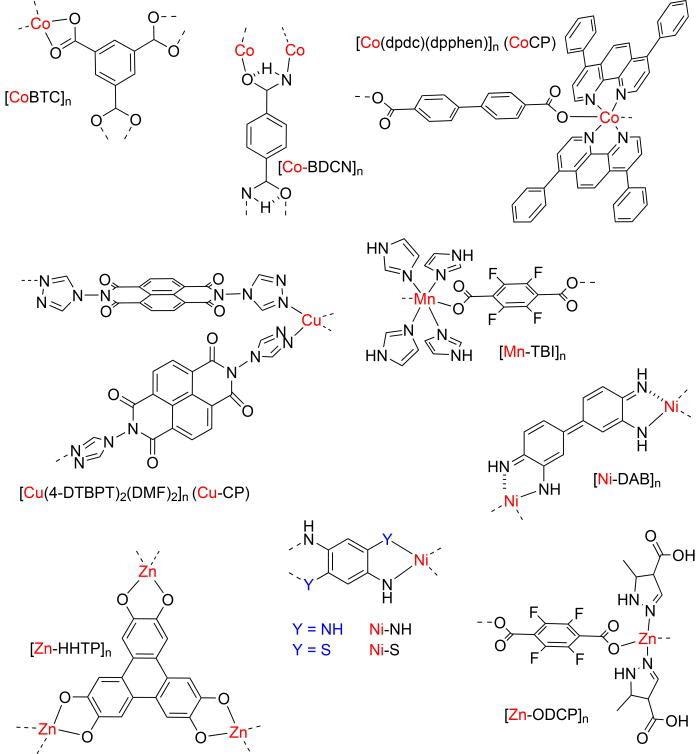
Chemical structures of selected CPs for battery applications. Image Credit: G. H. Morritt et al., Chem. Phys. Rev.
Not only are these similar in terms of composition to their organic and inorganic counterparts, but they also offer many of the beneficial properties of each type of linker. This has helped simplify coordination polymers’ manufacturing processes, often making these more energy-efficient and cost-effective.
Perhaps most notably, the properties of coordination polymers make these highly conductive, even rivaling the performance of inorganic semiconductors in terms of electrical conductivity.
The paper focuses on current research efforts into the use of coordination polymers in molecular electronics applications, providing valuable and detailed insight into these materials' design, processing, and properties. A number of application examples are also provided, including coordination polymers in sensor, battery, and photovoltaic environments.
Electronics are already highly prevalent throughout society, industry, medicine, and academia; and electronics are seeing ever-more widespread adoption in advanced applications such as light-emitting devices, sensor networks, charge storage, wearable displays, and more.
These increasingly demanding and varied use cases require the development of adaptable, scalable, and high-performance materials to meet a diverse array of application needs while – most crucially – ensuring that technology progresses with no adverse environmental impact.
The paper’s authors expect molecular electronics to play a central role in the ongoing drive to wholly sustainable devices. The tunability, solution processability, and strong electronic characteristics of coordination polymers lead the way in this area.
Further work into the effective control of coordination polymers’ morphological and electronic properties is therefore required, alongside the development of new fabrication techniques better able to leverage and accommodate these properties.
This will require more focus on the production of the materials in a device, and the device itself – an area which the authors point out has historically been an afterthought in many sectors.
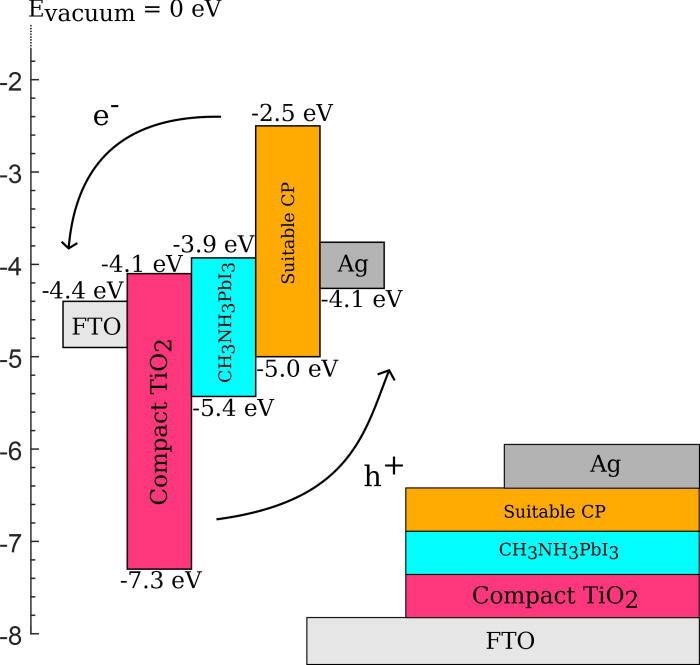
Schematic of a coordination polymer being employed as a hole transport layer in a perovskite solar cell. Typical values of conduction band and valence band energies are provided to demonstrate suitable energy level alignment to allow the flow of holes and electrons through the cell. Image Credit: G. H. Morritt et al., Chem. Phys. Rev.
One of the most promising properties of coordination polymers stems from the fact that they can be tuned through established synthetic chemistry techniques rather than relying on the implementation of large-scale and energy-intensive industrial processes and specialist disciplines.
This not only makes coordination polymers more cost-effective in terms of adapting them to meet specific application needs, but it also means that the processes of tuning these could be more energy efficient, simpler, and more environmentally friendly.
As electronic devices continue to expand in terms of their complexity and centrality to people’s everyday lives, it is becoming increasingly urgent that novel, flexible materials are developed, refined, and brought into widespread production.
The authors anticipate molecular electronics will become much more prevalent as time progresses, with conductive coordination polymers proving to be one of the fundamental enabling technologies which will lead to these entering more mainstream use.
References
G. H. Morritt, H. Michaels, and M. Freitag, "Coordination polymers for emerging molecular devices,” Chem. Phys. Rev. 3, 011306 (2022) https://aip.scitation.org/doi/10.1063/5.0075283
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.