Titanium (Ti) and its alloys are widely utilized in orthopedic implants and fittings owing to their outstanding biocompatibility, mechanical qualities, and corrosion resistance. However, due to their low wear resistance and hardness, there is a strong demand for Ti and its alloys with better mechanical qualities and lifetime in order to make them more acceptable.
Silver (Ag) and copper (Cu) are two of the most commonly explored alloying elements for improving Ti's mechanical qualities (Cu). These ions, however, can be exceedingly damaging to the host cells at higher doses. As a result, finding a safer biocompatible element to alloy with Ti is critical.
Gold (Au) is another highly biocompatible element from the same family as Ag and Cu. The selective coating of implants is also advantageous in overcoming the stress shielding effect, which is a major cause of implant failure. Previous research on the Ti–Au material system has focused on its structural, electrical, and mechanical properties in bulk; however, no attempt has been made to examine the combined biocompatibility and mechanical hardness of this alloy in thin-film form.
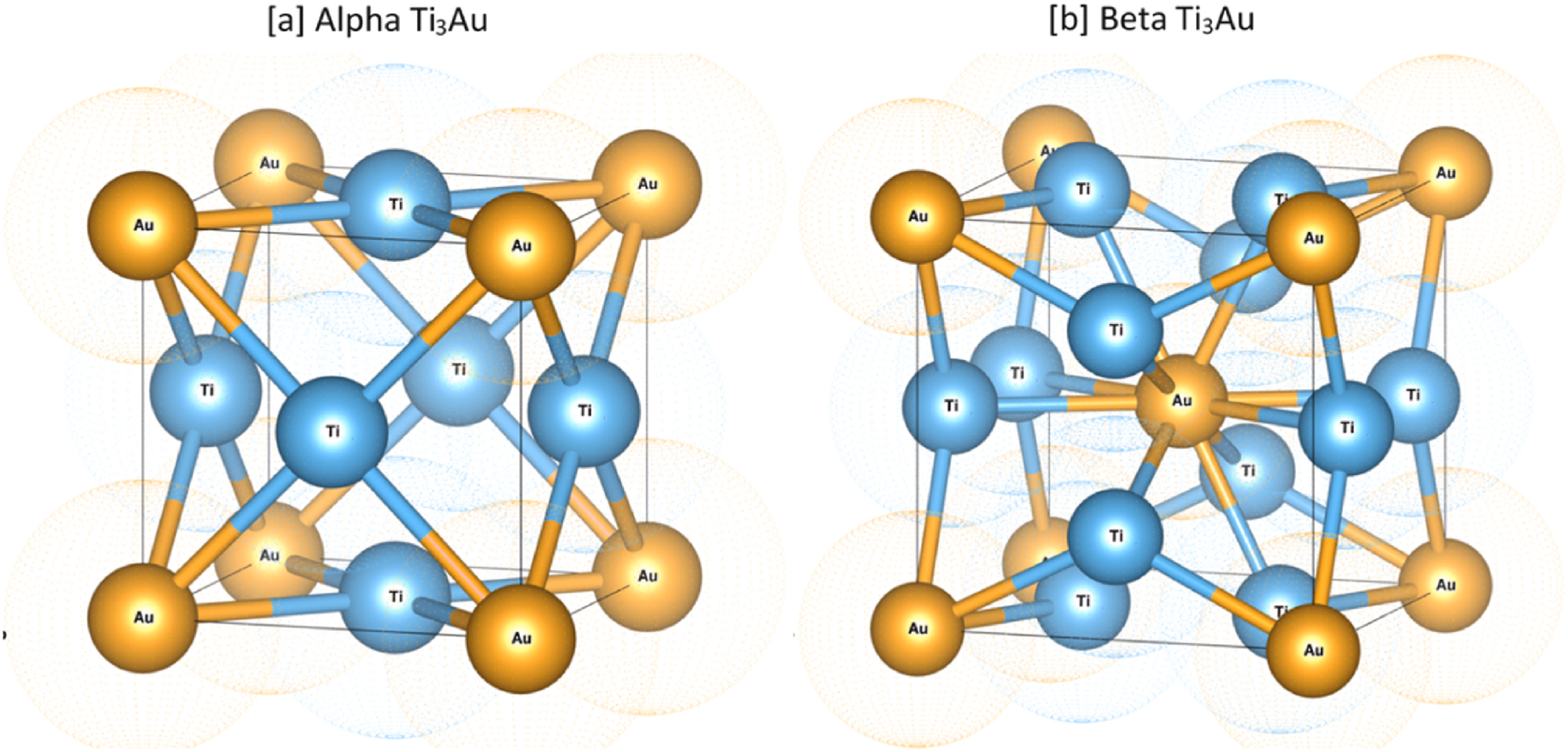
Unit cell structure showing the difference between the coordination configuration of Ti atoms in α and β-phase. Image Credit: Lukose, C et al., Bioactive Materials
About the Study
In the present study, the authors presented the development of thin films of Ti(1-x)Au(x) on Ti6Al4V and glass substrates by using magnetron sputtering in the whole x = 0–1 range. The impact of post-deposition heat-treatment versus in situ substrate heating on the biocompatibility and mechanical performance of Ti–Au films were investigated. The effect of covering the softer Ti6Al4V alloy with harder biocompatible thin films on the lifetime of orthopedic implants was also illustrated.
The impact of thermal activation on the surface morphology and elemental composition was also demonstrated, and the biocompatibility of all the prepared Ti-Au films was estimated.
An ex-situ heat treatment at 450 °C was used to explore the combined mechanical and biological properties of as-grown thin films of Ti–Au alloy in the composition range extending from pure Ti to pure Au in incremental steps of 15 at.% Au. In addition, the influence of in situ substrate heating on the quality of Ti–Au thin films were investigated, and the results were compared to films that have been heat-treated ex situ.
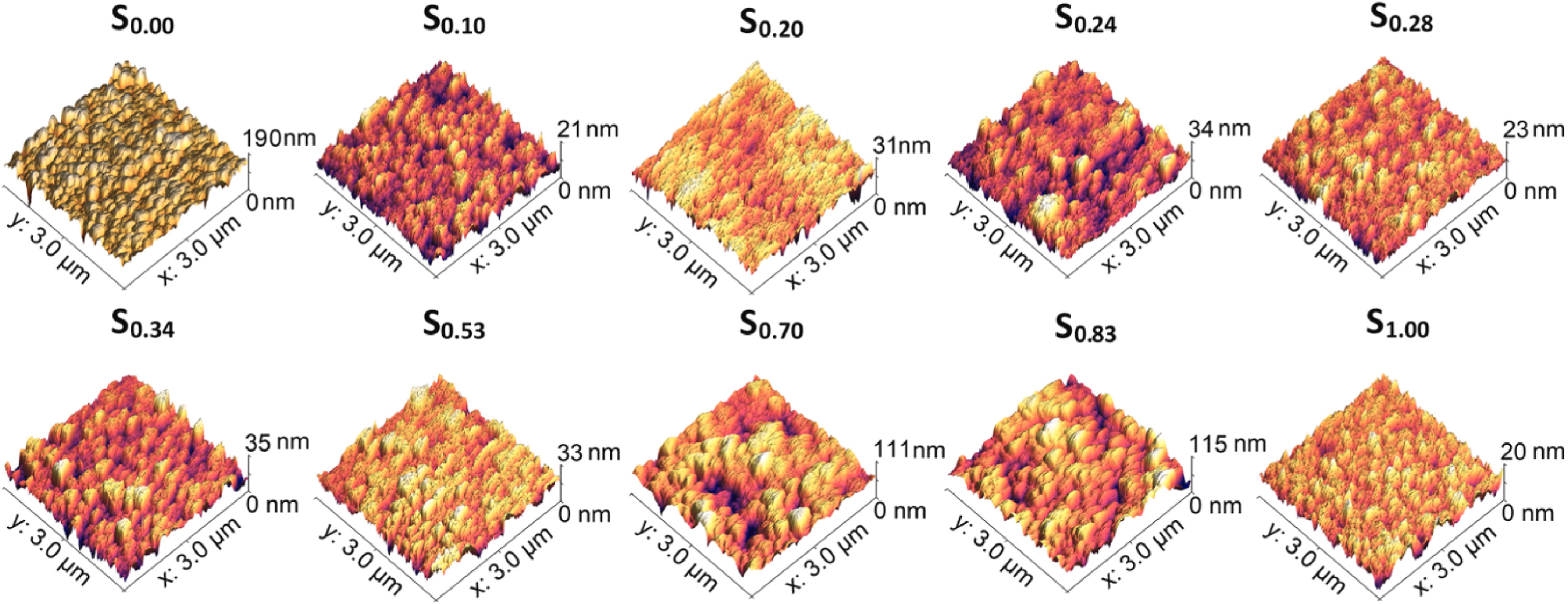
AFM micrographs of ex-situ heat treated Ti–Au thin films deposited on glass substrates with increasing Au composition. Image Credit: Lukose, C et al., Bioactive Materials
Observations
The mechanical characteristics of the Ti–Au alloy thin films were improved and attained a hardness value of more than 12 GPa along with an improvement in biological qualities of the individual Au and Ti elements. At normal temperature, Ti–Au thin films formed on glass or Ti6Al4V substrates had broad diffraction patterns covering the entire spectrum of peal positions of Ti–Au intermetallic. The polyhedral geometry of the as-developed Ti film transformed into a Ti–Au intermetallic surface having a fine texture and low surface roughness when Au was added.
Ti–Au films had a 250% increase in hardness compared to the bulk Ti6Al4V, and a 40% increment from 8.8 GPa to 12.3 and 11.9 GPa with ex situ and in situ heat treatment, respectively, which was correlated to changes in structural, chemical, and morphological properties. It was observed in X-ray diffraction studies that as-grown films were in nanocrystalline states corresponding to the Ti–Au intermetallic phase.
Following thermal activation, surface morphology photos demonstrated clear changes in grain shape, size, and surface roughness, while elemental analysis showed that in-situ substrate heating was superior for the production of oxide-free β-phases of Ti3Au. All Ti–Au films examined were non-cytotoxic to L929 mouse fibroblast cells, and their biocompatibility was confirmed by extremely low leached ion concentrations.
Thin films formed with a substrate temperature of 450 °C in situ had a better quality of crystallization of the Ti3Au intermetallic α and β-phases.
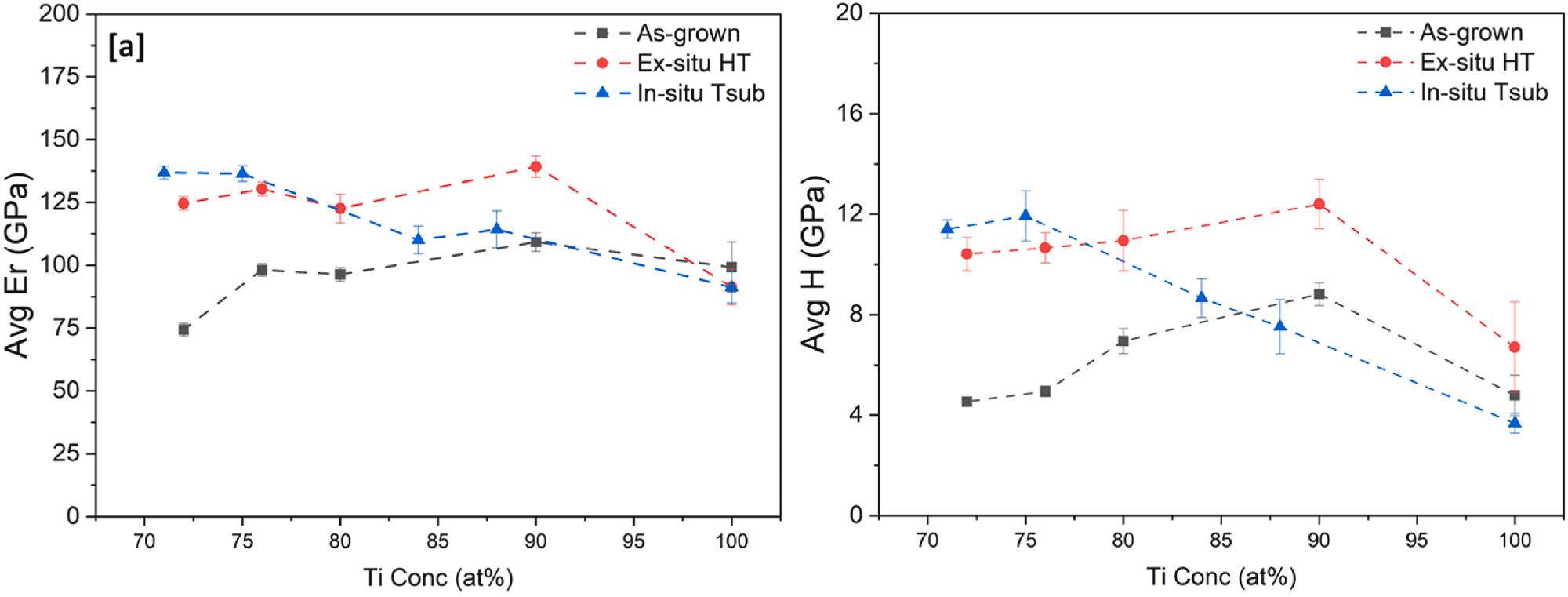
Variation of (a) Reduced elastic modulus (Er) and (b) Hardness (H) of Ti–Au thin films with increasing Au composition, deposited on glass substrates with in-situ substrate temperature compared with results from as-grown and ex-situ heat treated samples. Image Credit: Lukose, C et al., Bioactive Materials
Conclusions
In conclusion, this study demonstrated a straightforward single-stage scalable approach for producing Ti3Au thin films with an enhanced biological and mechanical performance by obtaining control over crystallization quality and introducing interstitial species.
Furthermore, in situ heat treatment offered a considerably simpler single-stage production process for producing good quality Ti–Au films and avoiding surface oxidation. Also, all the Ti–Au thin films produced in this study were found to be extremely biocompatible and noncytotoxic in L929 mouse fibroblast cells. Even after a longer extraction time of 168 hours, viability levels after incubations with all the prepared Ti–Au films were found in the range from 80 to 100% compared to untreated cells, while a number of samples had the potential to further increase L929 cell proliferation rates.
Overall, the authors believe that Ti-Au films have the capability to be the next-generation, biocompatible, super-hard coating to enhance the lifetime of orthopedic implants.
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.