Interest in the use of cellulose as an alternative, green material for numerous commercial and industrial applications has been driven by its abundance, sustainability, non-toxicity, and biodegradability.
Cellulose has been explored as a raw material for applications such as paper making, pharmaceuticals, tissue engineering, biomaterials, textiles, biofuels, and cosmetics. This natural polysaccharide contains abundant carbon and hydrogen and is composed of hundreds or thousands of linked homopolymer chains of residues. With a tough and inflexible structure caused by the presence of numerous hydrogen bonds, cellulose is a promising source of many important industrial chemicals.
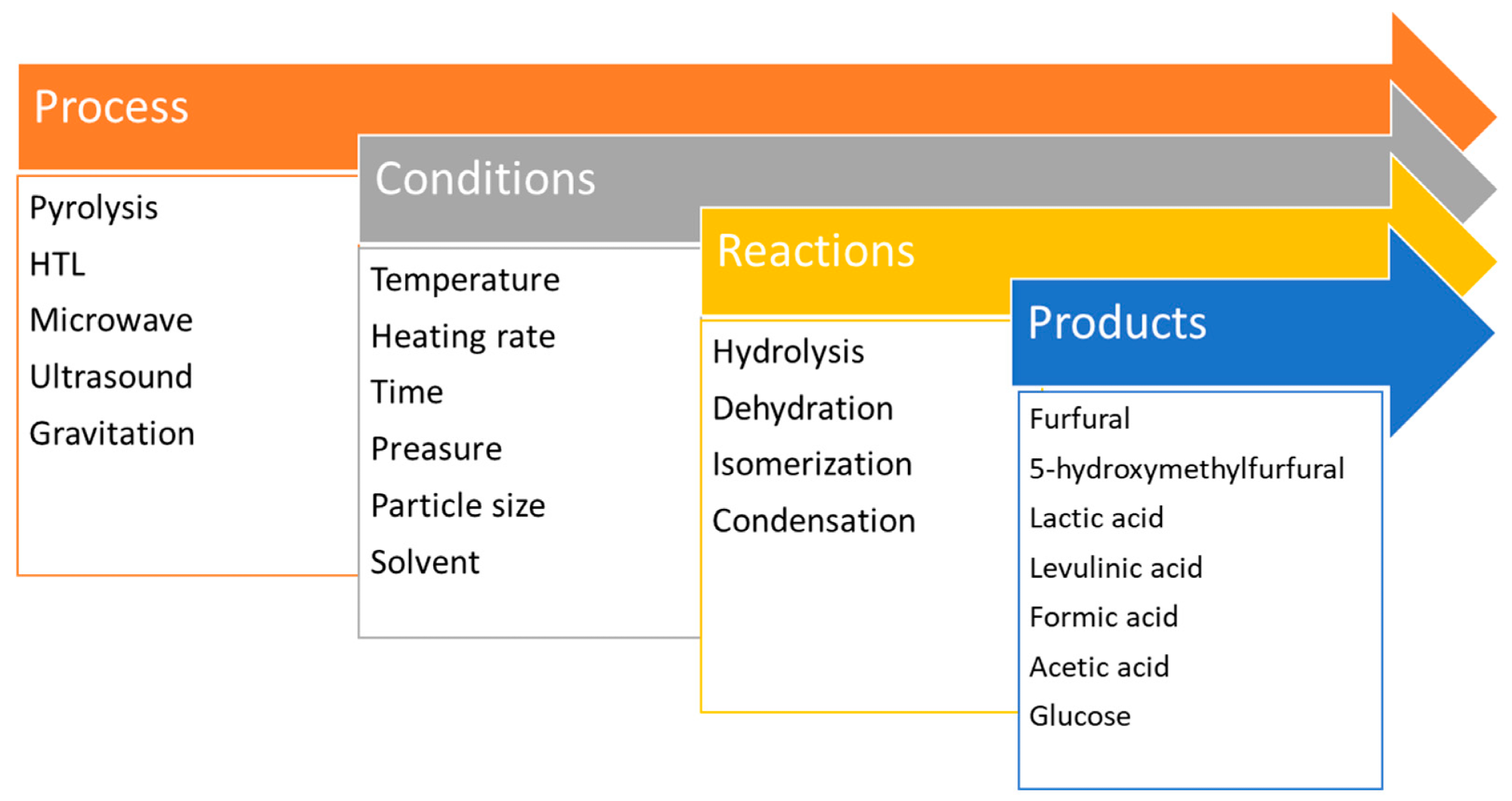
Schematic representation of biomass/cellulose liquefactions. Image Credit: Kryeziu, A et al., Polymers
Aerogels are one of the important industrial materials that can be produced from cellulose. To produce these materials, the rigid cellulose precursor must be converted to a liquid state. However, this process is complicated due to the insolubility of cellulose in common solvents such as water and organic solvents. Moreover, acids and enzymes can hydrolyze β-glycoside bonds.
Several methods have been explored recently to convert solid-state precursors into liquid-state materials. Early procedures involved the preparation of soluble derivatives such as acetate, cellulose nitrate, or xanthogenate. Other traditional methods include treating solid cellulose by dissolving it in solutions such as cuprammonium hydroxide or N-methylmorpholine-N-oxide.
Recently, research has focused on the development of new processes for liquifying cellulose. These include supercritical reactions using extremes of temperature and pressure, solvent mixtures, aggressive solvents, and using techniques such as ultrasonics, strong gravitational fields, and microwaves. The yield and composition of the final mixture are both strongly influenced by the preparation process and its parameters.
The Research
In the research, the authors have summarized the procedures for liquefying and dissolving rigid, insoluble cellulose to produce liquid mixtures containing the appropriate composition for the preparation of nanoporous carbonaceous materials. This is important for producing materials with appropriate porosity characteristics for use in commercial products such as carbon aerogels. The authors have reviewed 154 papers in the current literature.
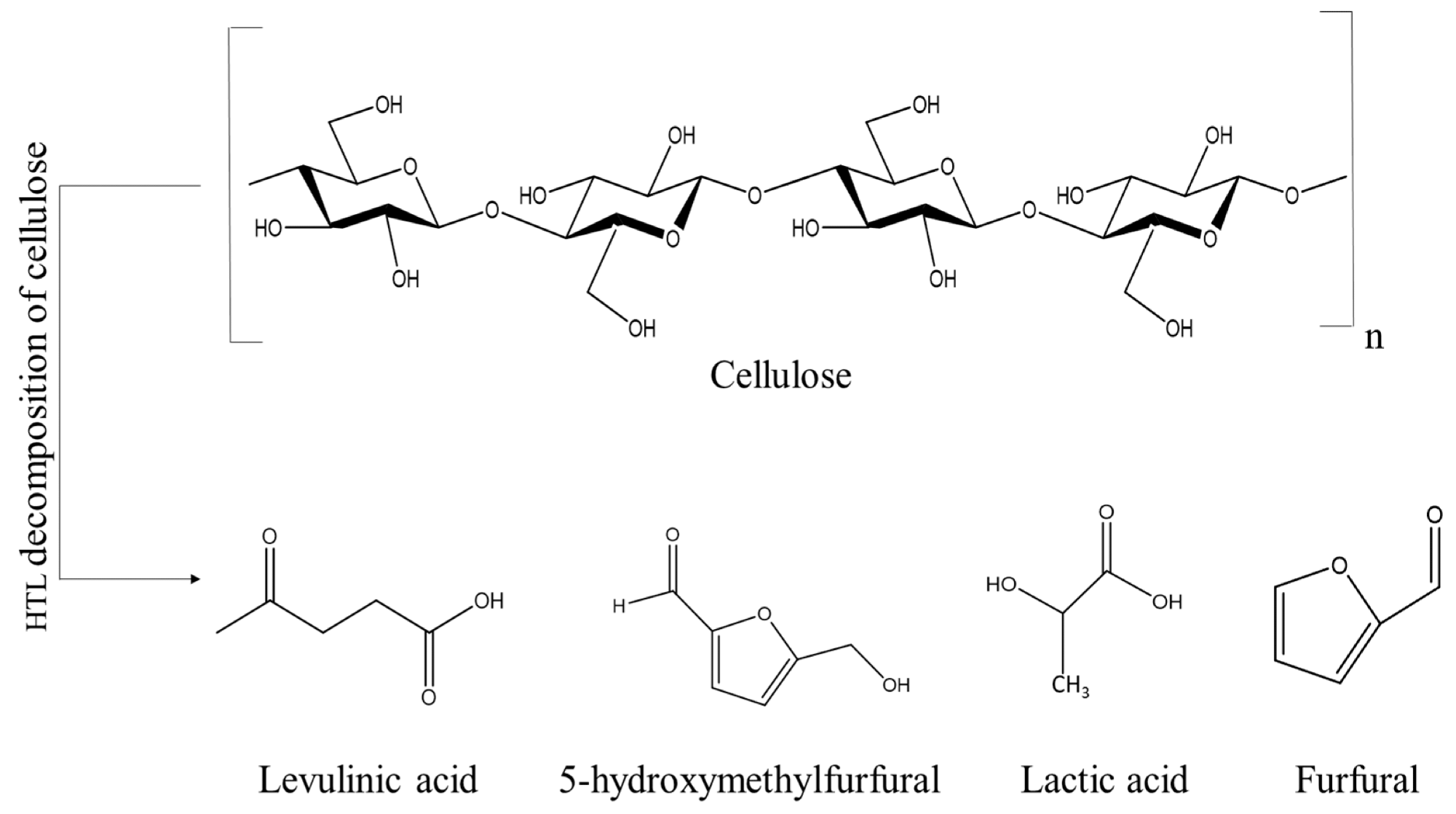
Decomposition products during cellulose liquefaction. Image Credit: Kryeziu, A et al., Polymers
Cellulose Decomposition Processes
Several processes have been developed based on the decomposition of cellulose, with the new study providing an in-depth analysis of these processes. Processes share some features but differ from each other in numerous ways.
Biomass pyrolysis is a commonly used process that produces bio-oil, char, and pyrolytic gases. Reaction parameters and the biomass feedstock are key factors in this complex process. Slow, fast, and rapid pyrolysis methods are all used, depending on the heating process.
In biomass pyrolysis, there are three processes: depolymerization, fragmentation, and char formation. The bio-oil formation is related to the decomposition of the biomass components and contains many complex compounds. It can undergo changes in physical properties and composition during storage.
Hydrothermal cellulose liquefaction is an alternative thermochemical process that is used to produce bio-oil from forestry and agricultural waste. Biomass decomposes in heated and pressurized organic solvents or water in the presence of catalysts. Processes can be either subcritical or supercritical. No prior drying is needed, and the highest yield is produced from biomass with high lignin content. Parameters and solvents influence the process and the properties and chemical composition of products.
Common Solvents Used
Other methods include microwave irradiation and the use of ultracentrifuge machines to aid gravity-induced decomposition and liquefaction. Cellulose dissolution is a complex process that requires chemical modification due to the presence of strong hydrogen bonds. Common solvents include alkalis, acids, metal salts, hydrated melted salts, organic solvents, and ionic liquids. Ideal solvents are hard to define as they are dependent on the purpose for which the cellulose needs to be dissolved.
Producing Tailored Carbon Materials from Cellulose
Direct carbonization is the simplest method to produce carbonaceous materials from cellulose precursors, but more advanced processes are required to produce materials with controlled properties, especially porosity. Methods based on liquid carbon precursors are the most attractive. Gelation, cross-linking, and templating can be used to produce carbon materials with well-defined structures, mechanical properties, and porosity.
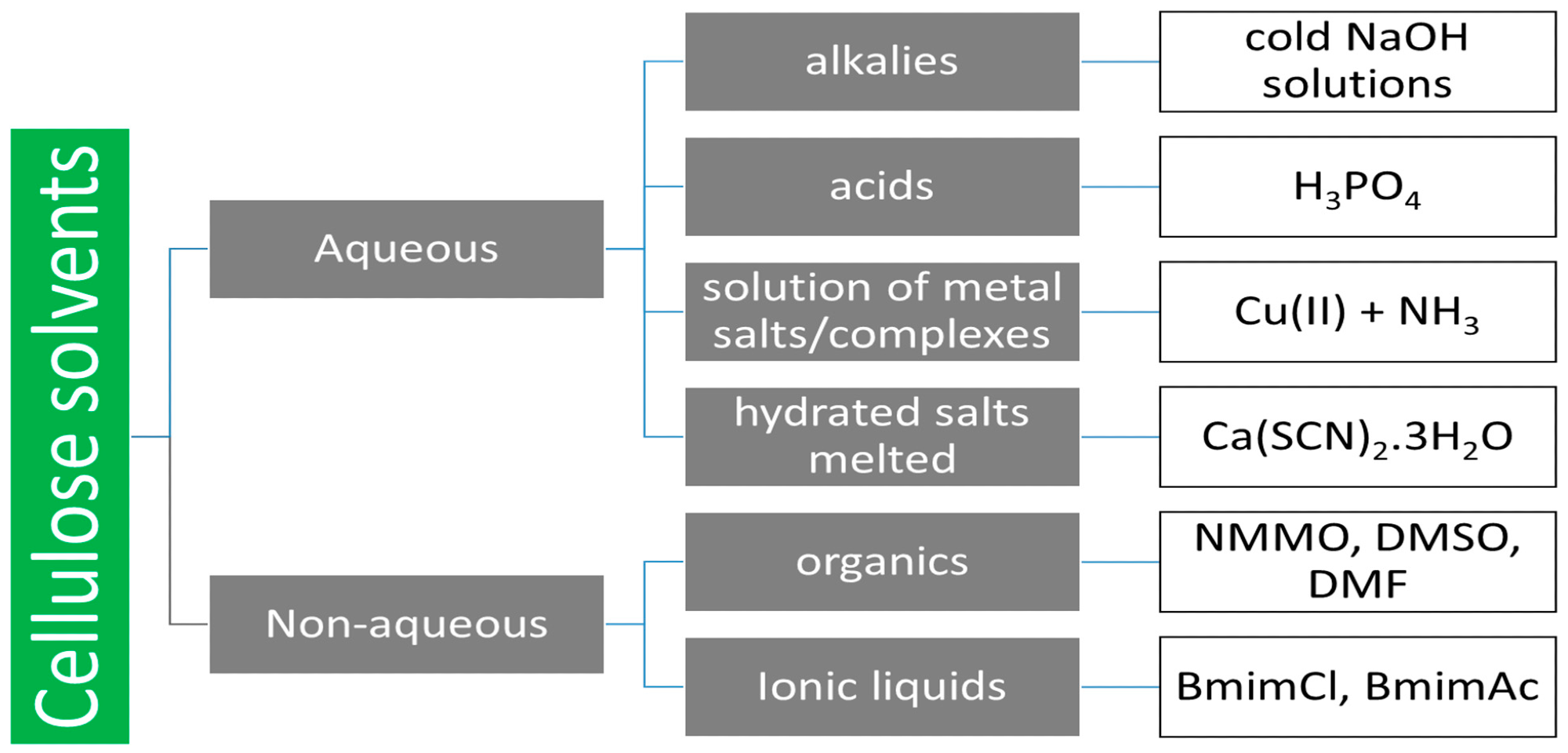
Cellulose solvents. Image Credit: Kryeziu, A et al., Polymers
Summary and Future Perspectives
The review has provided an in-depth summary of processes to prepare cellulose for use as a precursor for commercially important, tailored carbonaceous materials. Future perspectives highlighted by the authors include improving the uniformity and chemical stability of bio-oils to produce nanostructured carbon materials, optimizing processes to improve yield and avoid carbon loss, and research into producing carbon monoliths from biomass and cellulose with hierarchical porosity.
Further Reading
Kryeziu, A, Slovák, V & Parchaňska, A (2022) Liquefaction of Cellulose for Production of Advanced Porous Carbon Materials Polymers 14(8) 1621 | mdpi.com. Available at: https://www.mdpi.com/2073-4360/14/8/1621
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.