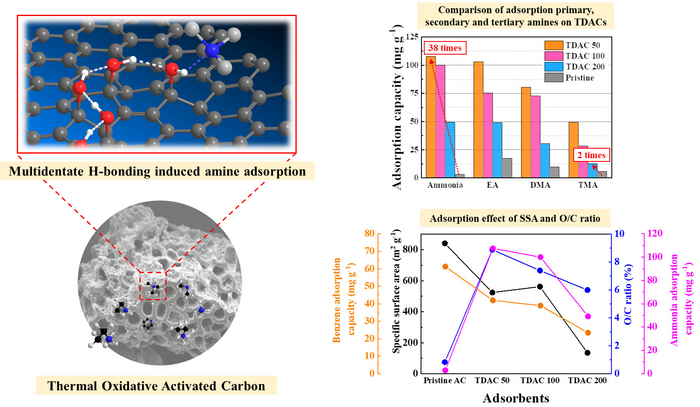
Adsorption mechanisms of heat-dried activated carbon and adsorption performance of nitrogen-containing odorous compounds. Image Credit: Korea Institute of Science and Technology
Conventional odor removal techniques use activated carbon as an adsorbent. However, activated carbon has a low level of recyclability, meaning that it is hard to remove the sources of complex odorous gases for reuse.
The removal of four representative nitrogen-containing odorous compounds (NOCs) from the air — ammonia, ethylamine, dimethylamine, and triethylamine — is significantly improved by an activated carbon manufacturing technology, according to a research team led by Dr. Jiwon Lee and Youngtak Oh of the Sustainable Environment Research Center at the Korea Institute of Science and Technology.
The study team not only increased the effectiveness of activated carbon for adsorbing odor-causing substances, but they also uncovered the process of adsorption between adsorbents and odorous gases, paving the way for the creation of a wider range of adsorbents for complicated odor substances.
After oxidizing the activated carbon with nitric acid, the study team could accurately adjust the surface oxidation level to boost the adsorption effectiveness of NOCs via a thermal annealing procedure.
They also discovered that the most oxidized heat-treated activated carbon could increase the removal efficiency of odor substances by up to 38 times compared to conventional activated carbon.
For the first time, the researchers were able to show that the oxygen atoms on the surface of oxidized activated carbon form strong hydrogen bonds with the amines in nitrogen-containing odor molecules.
This result validates the hypothesis that the adsorption impact of NOCs can be maximized by increasing the degree of oxidation so that more hydrogen bonds with amines can form on the surface of activated carbon.
Additionally, the study group showed that, in contrast to conventional gas reactions, the interaction between the adsorbent and odor substances was predominantly driven by the number of hydrogen bonds rather than proton affinity.
Furthermore, it was shown that thermally dried activated carbon (TDAC) increased trimethylamine selectivity by 13-fold. This result represents a significant improvement, as trimethylamine has the lowest adsorption efficiency among conventional NOCs.
Trimethylamine, a regulated odor substance in Korea, is a source of odors in landfills, agriculture, and sewage and wastewater treatment plants.
Heat-dried activated carbon, in particular, has an average recyclability of 93.8% for trimethylamine, exhibiting significant economic efficiency when compared to standard activated carbon’s recyclability of 63%.
By identifying the adsorption mechanism of odorous gases, we can develop materials that are specialized for removing specific gases, and heat-dried activated carbon, which undergoes an oxidation process, is relatively simple to produce and can be reused. Thus, it can be applied as a material for purification devices such as filters and masks.
Dr. Jiwon Lee, Sustainable Environment Research Center, Korea Institute of Science and Technology
This study was carried out as part of the KIST Major Projects funded by the Ministry of Science and ICT, and the findings were published in the most recent edition of the Journal of Cleaner Production (IF: 11.072, top 8.423% in JCR), an international journal in the field of environmental science.
Journal Reference:
Phyo, S., et al. (2023) Adsorption enhancement of hazardous odor gas using controlled thermal oxidation of activated carbon. Journal of Cleaner Production. doi:10.1016/j.jclepro.2023.136261.