Synthetic hydroxyapatite (HA) is of importance as a biomaterial as it is chemically similar to the mineral component of mammalian bone. As such the human body (and for that matter, the bodies of other mammals as well) is quite happy to integrate it into it.
It is one of few materials that are classed as bioactive, meaning that it will form strong chemical bonds with surrounding bone, unlike other materials such as alumina and zirconia, which are identified as foreign materials and become encapsulated by fibrous tissue.
HA is sometimes used in powder form, but in most cases it is used as a bulk material. To produce bulk samples generally requires requires sintering at elevated temperatures, usually in excess of 1000°C. High temperature exposure is often required in the production of HA coatings, another common application of the material.
Thermal Stability
Dehydroxylation
HA is a hydrated calcium phosphate material. It begins to dehydroxylate at about 800°Cto form oxyhydroxyapatite, or Ca10(PO4)6(OH)2-2xOxx, where =vacancy. This process is gradual and takes place over a range of temperatures.
Decomposition
Furthermore, HA also decomposes to form other calcium phosphates at elevated temperatures. Two mechanisms have been proposed for the decomposition as follows:
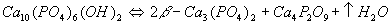
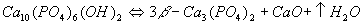
Of these, the former is the more accepted mechanism.
Further heating results in the transformation of β-tricalcium phosphate (β-Ca3(PO4)2), forming α-tricalcium phosphate. This usually requires exposure to temperatures in excess of 1350°C for this phase transformation to take place.
Regardless of which mechanism takes place, both result in the formation of soluble or resorbable calcium phosphates, which dissolve when exposed to physiological environments.
Factors Affecting Decomposition of Hydroxyapatite
Sintering Atmosphere
Under ambient sintering conditions decomposition has been reported to begin in the range 1050-1100°C, however, some materials have been observed to be resistant to decomposition until 1200°C.
Since a gaseous species exists on the products side of the decomposition reactions, sintering atmosphere would be expected to influence the decomposition kinetics of HA. Consequently, sintering under vacuum conditions can induce decomposition at lower temperatures, favouring the formation of water vapour. On the other hand sintering in a moist atmosphere can counteract this effect and delay decomposition to some degree.
Ca/P Ratio of Hydroxyapatite
HA has a theoretical Ca/P ratio of 1.67, however this is known to vary due to the HA structure’s willingness to undergo substitutions. Materials with Ca/P ratios different from 1.67 are known to be less thermally stable compared to stoichiometric HA.
Powder Synthesis Method
Some authors have reported that the method used to synthesise the HA powder can influence its thermal stability. They believe that HA powders synthesised using hydrothermal methods are more stable compared to precipitated powders. This may be attributed to the presence of adsorbed HPO42- ions.
Secondary Phases
Several people have tried reinforcing HA with secondary phases to improve its mechanical properties, which are not generally accepted to be suitable for load-bearing biomedical applications. In almost all cases, the presence of these phases has reduced the temperature at which decomposition begins. The same can be said for impurities in HA powders.
Primary author: Dr. Cameron Chai and Prof. Besim Ben-Nissan
Source: International Ceramic Monographs, Vol. 1, no. 1, pp. 79-85, 1994