Introduction
Photocatalytic water splitting is an attractive reaction in recent years for the photon energy conversion. These researches have been especially focusing on the visible light responsive materials from the point of view of solar energy. To date, a considerable visible-light-responsive photocatalysts have been produced, including metal (oxy) nitrides [1-5], metal (oxy) sulfides [6-7], metal sulfides solid solution [8-9], and RuO2-loaded α-Ge3N4 [10]. Among them, metal oxide materials based on Ti [11-12], Nb [13-14], Ta [15-17], Ga [18], In [19-20] and Sn [21] have been studied extensively as efficient photocatalysts under visible light irradiation. The feature of these metal oxide photocatalysts is to be composed of transition-metal cations with a d0 electronic configuration (Ti4+, Nb5+and Ta5+) or typical metal cations with d10 electronic configuration ( In3+, Ga3+ and Sn4+). In these compounds, the valence bands were mainly composed of the O-2p orbitals, whereas the conduction bands were formed by hybridzation of empty d or sp orbitals. The dispersion of the conduction band resulted in the mobility of photoexcited electrons, which was regarded as the main reason for high photocatalytic behaviors. Therefore, it is indispensable to control the energy structure for the development of visible-light-driven photocatalytic materials.
On the other hand, BiVO4 is an excellent photocatalyst for O2 evolution from aqueous AgNO3 solution under visible light irradiation because it has the mixed Bi-6s and O-2p valence band, which results in its high activity for photocatalytic O2 evolution [22-23]. In addition, bismuth-copper vanadate, BiCu2VO6 also has been prepared and it shows higher activity than BiVO4 [24]. Therefore, according to the guideline of energy band modulation, we designed and prepared bismuth-copper phosphate material, BiCu2PO6, which is effective for O2 evolution under visible light illumination.
Experimental Procedure
BiCu2PO6 was prepared by a high temperature solid reaction. In the synthesis of BiCu2PO6 powder, bismuth oxide (Nacalai Tesque, Inc., 99.9% pure), copper oxide (Nacalai Tesque, Inc.) and diammonium hydrogenphosphate (Kanto Chemical Co., Inc., 99.9% pure) in a 1:4:2 molar ratio were mixed by grinding in a agate mortar and heated at 573 K for 5 h, 773 K for 12 h, then 1023 K for 12 h. A dark green powder was obtained. The formation of BiCu2PO6 was confirmed based on X-ray diffraction (XRD) peak patterns reported previously [25]. The XRD pattern of the as-prepared material showed that BiCu2PO6 is in a single-crystalline phase, and belongs to orthorhombic system, with cell parameters, a = 11.776 Å, b = 5.173 Å, and c = 7.7903 Å. The morphology of the powders was observed by a scanning electron microscope (SEM).
Photocatalytic O2 evolution experiments were carried out in a reaction cell made of Pyrex glass, connected to a closed gas circulation system. The photocatalyst powder (0.5 g) was dispersed with magnetic stirrer into an aqueous solution (40 ml) containing 0.05 M AgNO3, as a visible light source 350 W Xe lamp combined with a cut-off filter (L-42, λ> 420 nm) was used. The amount of O2 evolved was determined with a gas chromatograph (Shimadzu, GC-8A, TCD, Ar carrier) directly connected to the closed gas circulation system.
Results and Discussion
Diffuse reflection spectrum of as-prepared BiCu2PO6 was measured using a UV-VIS-NIR spectrometer (Shimadzu, UV-3150) and was converted from reflectance to absorbance by the Kubelka-Munk method. BiCu2PO6 has two strong and broad absorption peaks at ca. 200-480 nm and 500–1000 nm, as shown in Figure 1. The strong absorption at ca. 200-480 nm has no steep absorption edge, and it extends to about 510 nm, which corresponds to the band-gap energy of 2.5 eV. The valence band of BiCu2PO6 consists of Bi-6s orbital and O-2p, and the rising of valance band level of O-2p resulting in a decrease of band gap. The inset is the diffuse reflection spectrum of BiVO4, which has strong and broad absorption in the region from 250 to 550 nm. It is different from the spectrum of the as-prepared BiCu2PO6. The reason is probably that bismuth and copper mixed together and the electronic state of phosphate was different from those of the vanadate.
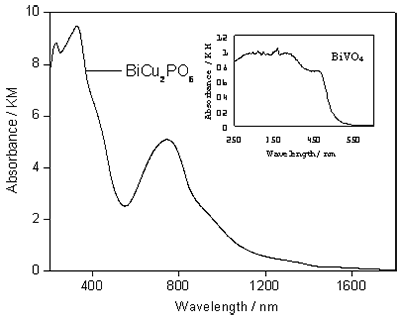
Figure 1. Diffuse reflectance UV-Vis absorption spectrum for the as-prepared BiCu2PO6 powder. Inset: diffuse reflectance UV-Vis absorption spectrum for BiVO4.
The morphology of BiCu2PO6 powder was evaluated by SEM observation (Figure 2). The SEM image of BiCu2PO6 showed that irregular particles were present, and that the estimated biggest size of particles was ca. 10 μm. The irregularity of the shape of the powders is caused by the defect of synthesis method of high temperature solid reaction itself.

Figure 2. SEM image of the as-prepared BiCu2PO6 powder.
Before irradiation, the suspension was stirred in the dark for 60 min to obtain a good dispersion and establish an adsorption–desorption equilibrium between the water molecule and the surface of catalyst. Figure 3 compares the result of photocatalytic oxygen evolution from aqueous suspension of BiVO4 (Afla Aesar, 99.9% pure), with that of BiCu2PO6 in the presence of Ag+ ions as sacrificial scavengers as a function of irradiation time. The initial rate of BiVO4 was about 2.5μmol/h, whereas that of BiCu2PO6 was only 0.8 μmol/h. This result indicated that BiCu2PO6 showed less activity than BiVO4 for photocatalytic O2 evolution under visible light irradiation.
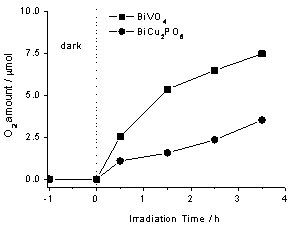
Figure 3. The amounts of evolved O2 from AgNO3 aqueous suspensions of as-prepared BiCu2PO6 and commercial BiVO4 as a function of the irradiation time
The lower activity for BiCu2PO6 than BiVO4 may be attributed to the difference of the composition of conduction band. As is known, the valance band of BiVO4 consists of the Bi-6s orbital mixed with O-2p, and the V-3d orbital forms the conduction band. Whereas for BiCu2PO6, the valance band is also composed of the Bi-6s orbital mixed with O-2p, but the Cu-3d orbital instead of V-3d consists of the conduction band. The different conduction band composition resulted in the wider band gap of BiCu2PO6 (2.5 eV) wider than that of BiVO4 (2.4 eV), and in the UV-vis diffuse reflection spectrum, BiCu2PO6 did not possess steep absorption edge in visible light region as BiVO4. Therefore, although BiCu2PO6 also has strong absorption in visible region and narrow band gap energy, its activity is less than that of BiVO4.
Conclusions
In conclusion, we observed O2 evolution from BiCu2PO6 under visible-light irradiation, although the activity was not significantly high. The present result suggests that a new series of compounds, metal bismuth-phosphates should have visible-light responsive potential. And this work also provides useful insight into the development of new photocatalysts for water splitting under visible-light illumination.
Acknowledgements
This research was supported by the 21st Century COE (Center of Excellence) Program from the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan in Nagaoka University of Technology, and by a CREST program from JST.
References
1. R. Asahi, T. Ohwaki, K. Aoki and Y. Taga, “Visible-Light Photocatalysis in Nitrogen-Doped Titanium Oxides”, Science, 293 (2001) 269-271.
2. M. Hara, J. Nunoshige, T. Takata, J. N. Kondo and K. Domen, “Unusual Enhancement of H2 Evolution by Ru on TaON Photocatalyst under Visible Light Irradiation”, Chem. Commun., 24 (2003) 3000-3001.
3. W. Chun, A. Ishikawa, H. Fujisawa, T. Takata, J. N. Kondo, M. Hara, M. Kawai, Y. Matsumoto and K. Domen, “Conduction and Valence Band Positions of Ta2O5, TaON, and Ta3N5 by UPS and Electrochemical Methods”, J. Phys.Chem. B, 107 (2003) 1798-1803.
4. K. Maeda, K. Teramura, T. Takata, M. Hara, N. Saito, K. Toda, Y. Inoue, H. Kobayashi and K. Domen, “Overall Water Splitting on (Ga1-xZnx)(N1-xOx) Solid Solution Photocatalyst: Relationship between Physical Properties and Photocatalytic Activity”, J. Phys. Chem. B , 109 (2005) 20504-20510.
5. K. Maeda, T. Takata, M. Hara, N. Saito, Y. Inoue, H. Kobayashi and K. Domen, “GaN:ZnO Solid Solution as a Photocatalyst for Visible-Light-Driven Overall Water Splitting”, J. Am. Chem. Soc., 127 (2005) 8286-8287.
A. Ishikawa, T. Takata, J. N. Kondo, M. Hara, H. Kobayashi and K. Domen, “Oxysulfide Sm2Ti2S2O5 as a Stable Photocatalyst for Water Oxidation and Reduction under Visible Light Irradiation (λ ≤ 650 nm)”, J. Am. Chem. Soc., 124 (2002) 13547-13553.
6. Ishikawa, T. Takata, T. Matsumura, J. N. Kondo, M. Hara, H. Kobayashi and K. Domen, “Oxysulfides Ln2Ti2S2O5 as Stable Photocatalysts for Water Oxidation and Reduction under Visible-Light Irradiation”, J. Phys. Chem. B, 108 (2004) 2637-2642.
7. Tsuji, H. Kato, H. Kobayashi and A. Kudo, “Photocatalytic H2 Evolution Reaction from Aqueous Solutions over Band Structure-Controlled (AgIn)xZn2(1-x)S2 Solid Solution Photocatalysts with Visible-Light Response and Their Surface Nanostructures”, J. Am. Chem. Soc., 126 (2004)13406-13413.
8. Tsuji, H. Kato and A. Kudo, “Visible-Light-Induced H2 Evolution from an Aqueous Solution Containing Sulfide and Sulfite over ZnS-CuInS2-AgInS2 Solid Solution Photocatalyst”, Angew. Chem. Int. Ed., 44 (2005) 3565-3568.
9. Sato, N. Saito, Y. Yamada, K. Maeda, T. Takata, J. N. Kondo, M. Hara, H. Kobayashi, K. Domen and Y. Inoue, “RuO2-Loaded -Ge3N4 as a Non-Oxide Photocatalyst for Overall Water Splitting”, J. Am. Chem. Soc., 127 (2005) 4150-4151.
10. Y. Inoue, M. Kohno, S. Ogura and K. Sato, “Dispersion of Ruthenium Oxide on Barium Titanates (Ba6Ti17O40, Ba4Ti13O30, BaTi4O9 and Ba2Ti9O20) and Photocatalytic Activity for Water Decomposition”, J. Chem. Soc. Faraday Trans., 84 (1998) 89-94.
11. T. Tabata, Y. Furumi, K. Shinohara, A. Tanaka, M. Hara, J. N. Kondo and K. Domen, “Photocatalytic decomposition of water on spontaneously hydrated layered perovskites”, Chem. Mater., 9 (1997)1063-1064.
12. Domen, J. N. Kondo, M. Hara and T. Takata, “Photo- and Mechano-Catalytic Overall Water Splitting Reactions to Form Hydrogen and Oxygen on Heterogeneous Catalysts”, Bull. Chem. Soc. Jpn., 73 (2000) 1307-1331.
A. Kudo, “Photocatalyst Materials for Water Splitting”, Catal. Surv. Asia, 7 (2003) 31-38.
13. H. Kato, K. Asakura and A. Kudo, “Highly Efficient Water Splitting into H2 and O2 over Lanthanum-Doped NaTaO3 Photocatalysts with High Crystallinity and Surface Nanostructure”, J. Am. Chem. Soc., 125 (2003) 3082-3089.
14. Machida, J. Yabunaka and T. Kijima, “Synthesis and Photocatalytic Property of Layered Perovskite Tantalates, RbLnTa2O7 (Ln = La, Pr, Nd, and Sm)”, Chem. Mater., 12 (2000) 812-817.
15. Shimizu, Y. Tsuji, M. Kawakami, K. Toda, T. Kodama, M. Sato and Y. Kitayama, “Photocatalytic Water Splitting over Spontaneously Hydrated Layered Tantalate A2SrTa2O7·nH2O (A=H, K, Rb)”, Chem. Lett., 31 (2002) 1158-1159.
16. Ikarashi, J. Sato, H. Kobayashi, N. Saito, H. Nishiyama and Y. Inoue, “Photocatalysis for Water Decomposition by RuO2-Dispersed ZnGa2O4 with d10 Configuration”, J. Phys. Chem. B, 106 (2002) 9048-9053.
17. J. Sato, N. Saito, H. Nishiyama and Y. Inoue, “Photocatalytic Activity for Water Decomposition of RuO2-Loaded SrIn2O4 with d10 Configuration”, Chem. Lett., 30 (2001) 868-869.
18. J. Sato, N. Saito, H. Nishiyama and Y. Inoue, “Photocatalytic Activity for Water Decomposition of Indates with Octahedrally Coordinated d10 Configuration. I. Influences of Preparation Conditions on Activity”, J. Phys. Chem. B, 107 (2003) 7965-7969.
19. J. Sato, N. Saito, H. Nishiyama and Y. Inoue, “New Photocatalyst Group for Water Decomposition of RuO2-Loaded p-Block Metal (In, Sn, and Sb) Oxides with d10 Configuration”, J. Phys. Chem. B, 105 (2001) 6061-6063.
A. Kudo, K. Omori and H. Kato, “A Novel Aqueous Process for Preparation of Crystal Form-Controlled and Highly Crystalline BiVO4 Powder from Layered Vanadates at Room Temperature and Its Photocatalytic and Photophysical Properties”, J. Am. Chem. Soc., 121 (1999) 11459-11467.
20. Sayama, A. Nomura, Z. Zou, R. Abe, Y. Abe and H. Arakawa, “Photoelectrochemical Decomposition of Water on Nanocrystalline BiVO4 Film Electrodes under Visible Light”, Chem. Commun., 23 (2003) 2908-2909.
21. H. Liu, R. Nakamura and Y. Nakato, “Bismuth–Copper Vanadate BiCu2VO6 as a Novel Photocatalyst for Efficient Visible-Light-Driven Oxygen Evolution”, Chem. Phys. Chem., 6 (2005) 2499-2502.
22. JCPDS Card No. 82-1312.
Contact Details
|