Jul 6 2015
Researchers at the Center for Self-Assembly and Complexity, Institute for Basic Science (IBS), Department of Chemistry and Division of Advanced Materials Science at Pohang University, South Korea have developed a new porous solid lithium ion battery (LIB) that has an improved performance and is free of risks associated with overheating.
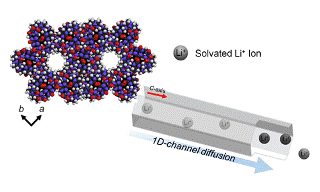 (Above) Simple incorporation of various lithium precursor to porous CB[6] exhibits high lithium ion conductivities, mobility and safer dried solid lithium electrolytes
(Above) Simple incorporation of various lithium precursor to porous CB[6] exhibits high lithium ion conductivities, mobility and safer dried solid lithium electrolytes
LIBs are efficient alternatives to the traditional lead acid batteries that have been used since the late 1850’s. LIBs have been mostly preferred in consumer electronics owing to their light weight, high energy density and minimized loss of charge when not being used. Lithium-ion cells carrying cobalt cathodes can store four-times the energy of lead-acid batteries and twice the energy of a nickel-based battery.
Although LIBs are considered as superior consumer batteries, they still have certain limitations. Existing manufacturing technologies have almost reached the theoretical energy density limit for LIBs while thermal runaway i.e. “venting with flame” due to overheating poses a major challenge.
US alone has reported nearly 40 incidents of fire or explosion due to LIBs used in consumer electronics since 2002. These batteries existing in different lithium-anode combinations and still remain as a vital component of modern consumer electronics irrespective of their poor track record at elevated temperatures.
The South Korean researchers have introduced a completely different approach. According to Dr. Kimoon Kim at IBS, “we have already investigated high and highly anisotropic [directionally dependent] proton conducting behaviors in porous CB[6] for fuel cell electrolytes. It is possible for this lithium ion conduction following porous CB[6] to be safer than existing solid lithium electrolyte -based organic-molecular porous-materials utilizing the simple soaking method.”
The existing LIB technology employs intercalated lithium with good performance. However, an increasing demand for a lighter and more powerful technology makes the study of novel electrolytes mandatory.
The newly developed battery is made up of pumpkin-shaped molecules called cucurbit[6]uril (CB[6]) arranged in a honeycomb-like structure. The molecules consist of an extremely thin 1D channel with an average diameter of 7.5Å [a single lithium ion is 0.76 Å, or .76 x 10-10 m] passing between them. Further, the physical structure of the porous CB[6] allows diffusion of lithium ions to battery more freely than in traditional LIBs, and avoids the need for separators found in other batteries.
The experimental results showed a favorable lithium ion conductivity of porous CB[6] solid electrolytes. To carry out a comparative analysis with the existing battery electrolytes, the research team performed a measurement of the lithium transference number (tLi+) that was observed at 0.7-0.8 over 0.2-0.5 of existing electrolytes.
In addition, they exposed the new batteries to high temperatures of up to 373K (99.85°C) which is more than the 80°C standard high temperature limit for exiting LIBs. During the experiment, the batteries were cycled at temperatures ranging from 298K to 373K (24.85°C to 99.85°C) for four days. The results did not indicate any thermal runaway after each cycle and no or minor change in conductivity.