Feb 24 2017
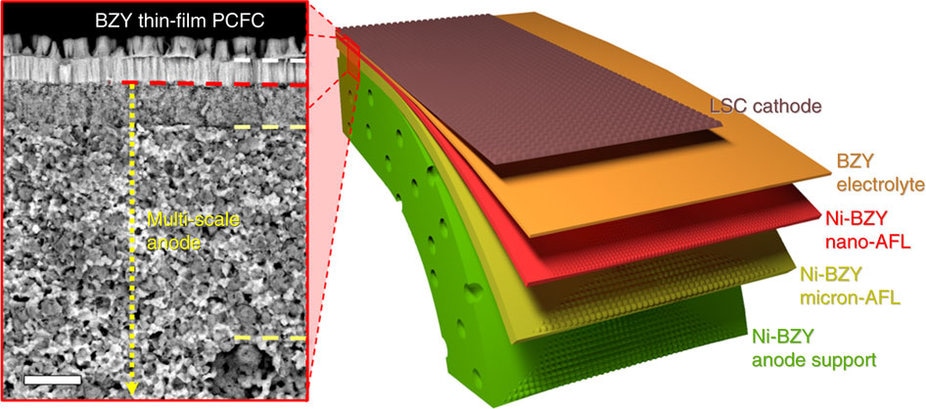 A schematic image of the proposed configuration of anode-supported PCFCs with thin-film BZY electrolytes along with a cross-sectional SEM image of the actually fabricated PCFC in the work. Scale bar, 5 μm. CREDIT: Nature Communications/Demonstrating the potential of yttrium-doped barium zirconate electrolyte for high-performance fuel cells
A schematic image of the proposed configuration of anode-supported PCFCs with thin-film BZY electrolytes along with a cross-sectional SEM image of the actually fabricated PCFC in the work. Scale bar, 5 μm. CREDIT: Nature Communications/Demonstrating the potential of yttrium-doped barium zirconate electrolyte for high-performance fuel cells
Fuel cells are devices that convert chemical energy from a fuel into electrical energy using an electrochemical reaction and can be used in stationary and portable power systems. Solid oxide fuel cells are one type of fuel cell with a range of applications that are characterized by the use of a solid oxide or ceramic material as the electrolyte.
Advantages of solid oxide fuel cells include high efficiency, high stability, fuel flexibility, low materials costs, and low emissions. However, solid oxide fuel cells require high operating temperatures in excess of 800 °C, which can increase both the running costs and the speed of degradation of the fuel cell.
Protonic ceramic fuel cells have been extensively studied as alternatives to solid oxide fuel cells due to their reduced operating temperatures (<600 °C), and low activation energies. However, they often face issues caused by poor chemical stability, resulting in poor long-term fuel cell stability.
Yttrium-doped barium zirconate (BZY) offers excellent chemical stability and high bulk conductivity but has thus far been limited in applications in protonic ceramics due to poor performance. Researchers have now achieved a technological breakthrough in this area and have produced a protonic ceramic fuel cell with a thin-film deposited BZY electrolyte and a record high power output.
Until now, protonic ceramic fuels developed with BZY electrolytes have been characterized by high ohmic resistance and low power outputs, caused by highly resistive grain boundaries. Previous attempts to reduce grain boundaries in BZY have resulted in the growth of large BZY grains with reduced grain boundary resistance, but it has not been possible to eliminate grain boundaries completely.
However, a team of researchers from Korea has now been able to produce a thin-film BZY electrolyte with no grain boundaries.
BZY electrolytes of protonic ceramic fuel cells are typically produced by thin-film deposition onto the anode material. The resulting structural characteristics of the BZY membrane are therefore dependent on the anode having a high-quality surface suitable for thin-film deposition.
The team from Korea utilized an optimized ‘multi-scale’ anode with reducing grain and pore sizes to grow a thin BZY electrolyte film. The multi-scale anode consisted of a nanostructured anode functional surface layer on a conventional powder-processed anode body.
The functional surface layer of the anode reduces defects and roughness of the anode surface, increases the boundary length, and increases the contact between the electrolyte and the anode, resulting in reduced interfacial resistance. The growth of the BZY electrolyte on the multi-scale anode resulted in a columnar microstructure and the elimination of grain boundaries, decreased membrane thickness, and increased bonding between the anode and electrolyte.
The structural improvements of the BZY electrolyte led to significant improvements in the performance of the resulting protonic ceramic fuel cells. Electrochemical characterization of the BZY protonic ceramic fuel cells showed record-breaking power outputs of 740 mW cm-2 at 600 °C, a significant improvement compared with the previously reported maximum power output of protonic ceramic fuel cells (650 mW cm-2 at 600 °C).
Furthermore, the ohmic and polarization area-specific resistances were significantly lower than previously reported protonic ceramic fuel cells, and the open-circuit voltages achieved were close to the theoretical value for BZY (~1.08 V at 600 °C).
This research represents a significant step towards the practical use of protonic ceramic fuel cells. The improvement in performance in BZY protonic ceramic fuel cells provides a solution to the challenge of low chemical stability faced by high performance protonic ceramic fuel cells thus far. Furthermore, continued optimization of the BZY protonic ceramic fuel cell, including optimization of the cathode material, could lead to further increased performance.
Source:
Bae K., Jang D.Y., Choi H.J., Kim D., Hong J, Kim B-K., Lee J-H., Son J-W., Shim J.H., Nature Communications, 2017, 8, 14553.