Dec 18 2017
Researchers from Ames Laboratory of the U.S. Department of Energy have found out an innovative technique for encasing metal below a single layer of graphite, which may result in new and better regulated characteristics of these kinds of materials.
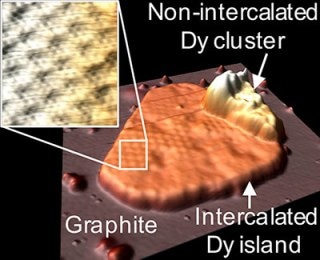 A “mesa” of dysprosium, draped with a single layer of graphene. Research from Ames Laboratory suggests that this method will work with a wide variety of metal and graphite combinations, opening up the possibility of diverse applications including quantum computing, solar cells, catalysis, and sensing.
A “mesa” of dysprosium, draped with a single layer of graphene. Research from Ames Laboratory suggests that this method will work with a wide variety of metal and graphite combinations, opening up the possibility of diverse applications including quantum computing, solar cells, catalysis, and sensing.
Graphene - two-dimensional form of graphite - was first synthesized in the year 2004, and due to the fact that electrons move swiftly along its surface, it has great potential as a highly useful material in a range of applications such as catalysis, quantum computing, sensing and solar cells. To enable graphene to satisfy that capability, fundamental discovery science at Ames Laboratory is working on mastering its fabrication in combination with other materials, which is a difficult and intricate procedure carried out at the atomic scale in ultra-high vacuum lab conditions.
The team enclosed dysprosium (a magnetic rare-earth metal) by striking bulk graphite’s top layer with ions to make flaws on its surface, and then performed high-temperature deposition of the metal. The resulting material was “mesas” or islands of dysprosium found below a single graphene layer. The formations are considerably different from the materials that the two-dimensional materials experts at the Laboratory have come across thus far.
It’s well-known that certain metals can be embedded between bulk graphite layers. But these mesas form at the top graphite surface only, and they are pure metal composed of multilayers, which is a first. The combined properties of the metal plus graphene may be very different than other, previously produced materials. That’s something we’re exploring now.
Ann Lii-Rosales, Research Assistant
The team could also accomplish similar mesa-like formations with two transition metals, namely copper and ruthenium.
That suggests we have a very adaptable recipe for producing this kind of surface material, which makes its discovery very exciting in terms of potential applications. If we can control the process so that we can deliberately pattern the formation of these little metal slabs, perhaps we can harness and control their magnetic and electronic properties.
Pat Thiel, Scientist, Ames Laboratory and Distinguished Professor of Chemistry and Materials Science and Engineering, Iowa State University
Further discussion of the study can be seen in the paper “Defect-mediated, thermally-activated encapsulation of metals at the surface of graphite” published in the Carbon journal. Yinghui Zhou, Ann Lii-Rosales, Minsung Kim, Mark Wallingford, Dapeng Jing, Michael C. Tringides, Cai-Zhuang Wang, and P.A. Thiel are the authors of the paper.
The U.S. Department of Energy Office of Science primarily supported the study. The study was also partially supported by a grant of computer time at the National Energy Research Scientific Computing Centre (NERSC), a DOE Office of Science User Facility.
Ames Laboratory is a U.S. Department of Energy Office of Science national laboratory operated by Iowa State University. Ames Laboratory develops new materials, energy solutions and technologies. We put to good use our expertise, distinctive potentials and interdisciplinary partnerships to overcome global challenges.
DOE’s Office of Science is the single largest supporter of fundamental research in the physical sciences in the United States, and is striving to address some of the most critical difficulties in the present day. For further information, please visit science.energy.gov.