Jul 10 2018
Researchers from the Ghent University have used computer simulations to discover a new material that efficiently changes methanol into building blocks for plastics and fuels. This conversion has the potential to substitute traditional petrochemical processes that are based on crude oil. The advantage of the methanol-to-olefins (MTO) process is that methanol can be created from any carbon-containing material paving the way for the utilization of CO2 captured from the atmosphere. Based on that view, the MTO process can play a vital role in lowering CO2 emissions and man’s reliance on fossil fuels.
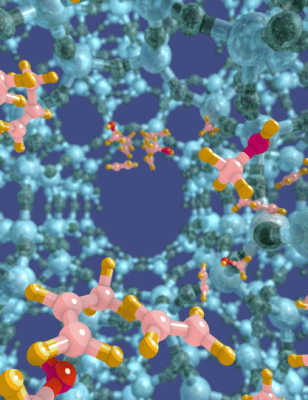 Credit: Ghent University
Credit: Ghent University
The MTO conversion takes place in a zeolite catalyst, which is a solid material having pores on the nanoscale. A big disadvantage of existing MTO catalysts is that they create plenty of unwanted side products and even deactivate after a brief period. By itself, the MTO process cannot contend with oil-based petrochemical processes that have been enhanced for many years. Presently, the MTO process is already commercialized in China but its low efficiency impedes large-scale applications around the world.
An international team of scientists recently discovered a clever way to improve the performance of MTO catalysts, resulting in a material that produces two times more of the most beneficial product and has nine-times longer lifetime compared to catalysts in the market. Therefore, the team adapted the catalyst’s architecture at the molecular level by adding more building blocks in the pores of the material.
This was facilitated by computer simulations of the material to discover its actions at the molecular level. The simulations were done at the Center for Molecular Modeling of Ghent University under the guidance of Prof. Veronique Van Speybroeck and Dr. Kristof De Wispelaere.
The new material was synthesized and examined on a lab scale by a large team of scientists at Delft University of Technology, King Abdullah University of Science & Technology, Utrecht University, Eindhoven University of Technology, University of Oslo and ETH Zurich. The exciting results provide design strategies for a new generation of materials that will influence the transition towards more sustainable chemical processes and will be reported in Nature Chemistry.