Jul 31 2018
Usually, it is quite difficult to synthesize the natural products terpenes in the laboratory. Now, chemists from the University of Basel have come up with an innovative method to synthesize these natural products that imitate nature.
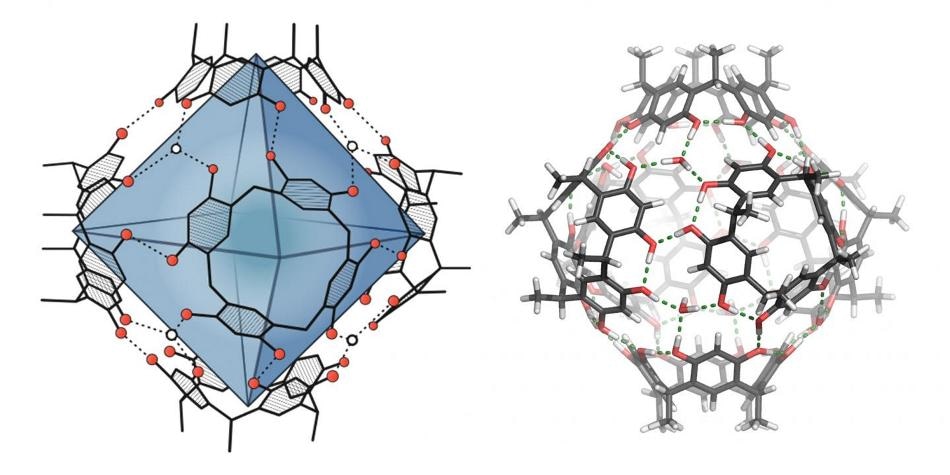 On the left, the around 1.4 cubic nanometer-large cavity within the molecular capsule is highlighted in blue. On the right, the cohesion of the capsule via hydrogen bonds (green dashed lines) is visible. (Image credit: Image: University of Basel, Department of Chemistry)
On the left, the around 1.4 cubic nanometer-large cavity within the molecular capsule is highlighted in blue. On the right, the cohesion of the capsule via hydrogen bonds (green dashed lines) is visible. (Image credit: Image: University of Basel, Department of Chemistry)
The decisive step occurs within a molecular capsule, which allows the reaction. The results of the study have been reported in the journal, Nature Catalysis.
Terpenes are known to be the largest group of chemical compounds to occur in nature. For instance, they include steroids, several essential oils, and clinically relevant substances including the chemotherapy medication paclitaxel or the antimalarial drug artemisinin.
Although increasingly refined synthesis techniques are available, chemists have found it extremely challenging to produce these structurally complex compounds in the laboratory. Most often, the process needs numerous but not invariably selective synthesis steps, and the yields are likely to be low.
Nature Points the Way
Headed by Professor Konrad Tiefenbacher from the University of Basel's Department of Chemistry, the team has now come up with a unique synthesis idea for terpenes replicated from nature.
This decisive step occurs in the cavity of a spherical compound – called a molecular capsule. While the resorcinarene capsule utilized has been known for approximately two decades, its catalytic effect has only been recently explained by Tiefenbacher and others. In the case of organic solvents, the resorcinarene capsule forms itself from six compounds, which are smaller and shaped like a ring, with the aid of hydrogen bonds.
In an analogous manner to nature, the research team headed by Tiefenbacher commences with a starting material - enclosed by the capsule - for the synthesis. The environment of the capsule subsequently allows the formation of terpene. Moreover, control elements that were earlier incorporated into the precursor help prevent unnecessary side effects and direct the change towards the preferred product.
Proven Applicability
The four-step synthesis of isolongifolene - a natural product - demonstrated the applicability of the concept, with the formation of a terpene compound shaped like a ring and catalyzed by the capsule as the main step. When compared to traditional syntheses of terpenes, this proved to be successful—in considerably lesser steps and also with a good yield.
The Basel chemists used labeled precursors and computer simulations to explain the reaction mechanism.
Our next goal is to use capsules as an artificial enzyme in the creation of even more complex terpenes. In order to do so, we must learn to better control the spatial arrangement of the precursor within the capsule, either by modifying the existing system or by developing new catalysts.
Professor Konrad Tiefenbacher
This can possibly pave the way for producing terpene compounds that may otherwise not be instantly accessible.