Drug development has become increasingly difficult and understanding formulation design space and the interplay raw materials properties and processing parameters have on the final drug product have never been more important.
The rise of computational methods for drug design has yielded increasingly complex molecules which, although very potent and target-specific, often have poor drugability. An increased emphasis on quality-by-design approaches to drug development has meant that the regulatory authorities, now more than ever, expect to see sound scientific rational behind drug design choices.
To address these issues, George Butcher (Pharmaceutical Product Manager) and Tim Smith (Applications Manager), from Renishaw’s UK Raman team, will be hosting a live webinar on the use of Raman imaging in pharmaceutical drug development.
The webinar, on Thursday 27th September at 3:00 pm (UK), 9:00 am (Chicago), will focus on how Raman imaging can help you to understand your formulation, troubleshoot formulation problems and de-risk the drug development process.
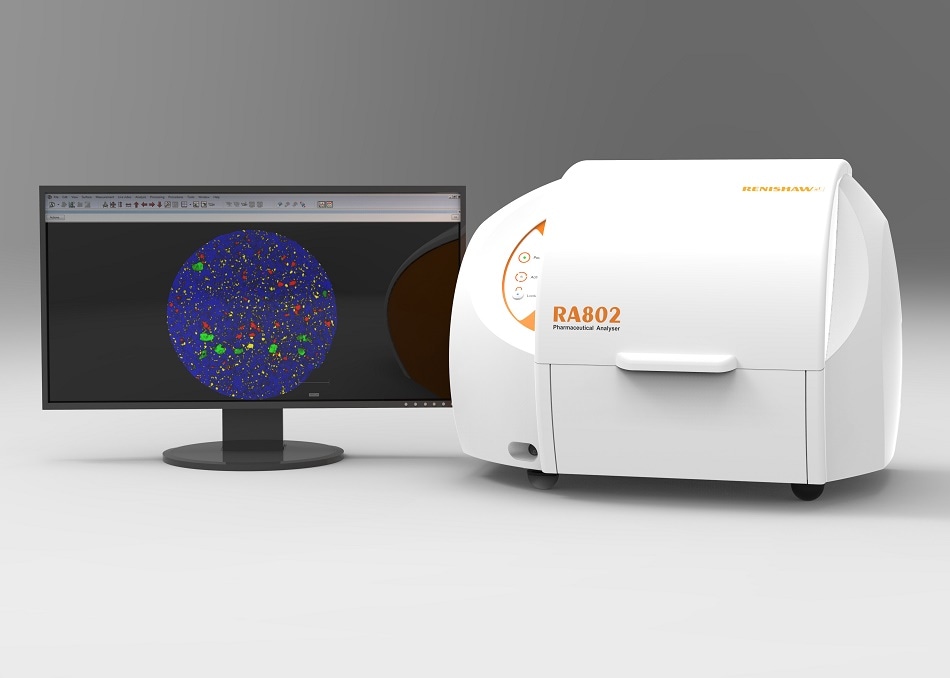
Renishaw’s RA802 Pharmaceutical Analyser enables the imaging of whole tablets using LiveTrack focus tracking technology and collects high quality spectral datasets for analysis. Find out how Renishaw’s Raman instruments can be used for pharmaceutical research by visiting www.renishaw.com/raman