Mar 25 2019
Tokyo Metropolitan University researchers have demonstrated that a recently engineered catalyst composed of gold nanoparticles reinforced on a metal oxide framework exhibits breakdown of ammonia contaminations in air, with outstanding selectivity for conversion to nitrogen gas. Significantly, it is operative at room temperature, making it ideal for daily-use air purification systems. The team positively discovered the mechanism behind this behavior, thus making way towards the design of other unique catalytic materials.
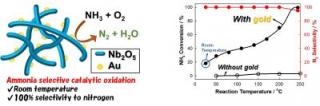 (left) Simplified schematic of gold nanoparticles attached to a niobium oxide framework. (right) Conversion efficiency of filter with and without gold, plus selectivity for conversion to nitrogen and water. The graph shows that frameworks loaded with gold nanoparticles show vastly improved activity at room temperature, with excellent selectivity over the entire temperature range. (Image credit: Toru Murayama)
(left) Simplified schematic of gold nanoparticles attached to a niobium oxide framework. (right) Conversion efficiency of filter with and without gold, plus selectivity for conversion to nitrogen and water. The graph shows that frameworks loaded with gold nanoparticles show vastly improved activity at room temperature, with excellent selectivity over the entire temperature range. (Image credit: Toru Murayama)
Many are acquainted with the characteristic, sharp odor of ammonia. It is a standard industrial chemical, mainly used as feedstock for fertilizers as well as disinfectants in both medical settings and household. It is also extremely poisonous when concentrated; the United States Occupational Safety and Hazard Administration has a stringent upper limit of 50 parts per million in breathing air averaged over an eight-hour working day and forty-hour work week. Given its broad industrial use and occurrence in nature, it is vital that actual measures are in place to eliminate undesirable ammonia from the atmosphere in human’s everyday working and living settings.
Catalysts, similar to the ones seen in the catalytic converters of cars, can aid in solving this issue. In contrast to filters which merely capture dangerous substances, catalytic filters can help break ammonia down into harmless products such as water and nitrogen gas. Not only is it harmless, preventing the accumulation of toxic chemicals, it also makes it needless to swap them regularly. However, presently existing catalysts for ammonia only work at temperatures of more than 200 °C, making them unproductive as well as unsuitable for household settings.
Currently, a team guided by Project Professor Toru Murayama from Tokyo Metropolitan University has built a catalytic filter which can operate at room temperature. Made up of gold nanoparticles stuck onto a structure of niobium oxide, the newly engineered filter is extremely selective in what it changes ammonia to, with virtually all conversion to harmless nitrogen gas and water and no nitrogen oxide derivatives. This is called selective catalytic oxidation (SCO). They partnered with industrial partners from NBC Meshtec Inc. to create a working model; the filter has already been applied to decrease gases tainted with ammonia to untraceable levels.
Significantly, the team also effectively discovered the mechanism by which the material functions. They demonstrated that gold nanoparticles have a key role to play, with increased loading resulting in higher catalytic activity; they also learned that the choice of framework was very important, exhibiting experimentally that chemical sites called Brønsted acid sites on the niobium oxide backbone played a vital role in how selective the material was. The researchers anticipate that basic design principles such as this may find application in the creation and alteration of other catalytic materials, spreading their increasing range of applications.
This research was aided by a grant from the Platform for Technology and Industry of the Tokyo Metropolitan Government.