According to the latest research, pollution and climate change are some of the biggest threats to the modern world. The fossil fuels which have been used for a long time resulted in the emission of byproducts that are severely harmful to the environment. Using biomass in batteries has the potential to mitigate some industrial impacts upon the environment.
Lithium-Sulfur Batteries
According to the study, because of the high efficiency, low self-discharge speed, and extended lifetime, lithium-based batteries are particularly appealing. The chemical reactions occur between a Li-metal anode and a sulfur-based cathode power Li–S batteries.
During battery depletion, the final reaction product is Li2S. In 2011, activated carbons (AC) produced from pig bones were used as sulfur hosts in the positive electrode of Li–S batteries for the first time. Activated carbons derived from agricultural leftovers were first utilized a few years later.
Activated Carbons (AC)
The amorphous non-graphite material and a suitable structure of standardized micro, meso, and macropores are termed Activated Carbon. The micro, meso, and macropore sizes are less than 2nm, 2-50nm, and more than 50nm, respectively. The rapid decomposition or combustion of carbon-based precursors generates AC.
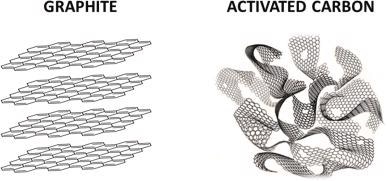
Illustrative representation of three-dimensional crystal lattice of graphite and amorphous activated carbon. Image Credit: Benítez, A et al., Renewable and Sustainable Energy Reviews
ACs contain carbon as the main constituent. From the standpoint of environmental contamination and minimal raw material accessibility, biomass precursors for AC production are the best option. The production of AC from natural biomass is commercially viable and has several advantages.
Biomass Implementation in Li-S batteries
The famous 5 types of biomass used in Li-S batteries are woody biomass, herbaceous and agricultural residues, aquatic, animal and human biomass, and contaminated and industrial biomass wastes.
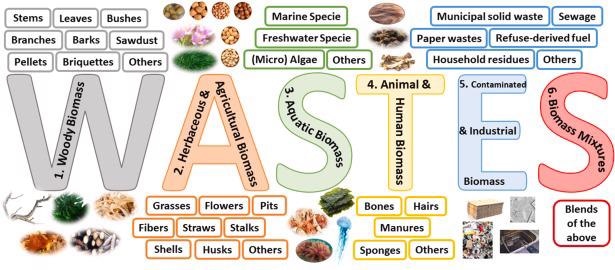
Classification of the different sources of biomass for energy according to their origin. Image Credit: Benítez, A et al., Renewable and Sustainable Energy Reviews
Woody Biomass
According to the latest research, OH and ZnCl2 were utilized for activation. Temperatures of carbonization vary as well, ranging from 700 to 1000 degrees Celsius. The fact that non-activated carbons have much lower SBET values than activated carbons appears fair. Xu et al. published on the utilization of activated carbons from forest wastes as cathodic matrices in Li–S batteries.
Agricultural Biomass
The agricultural residues analyzed were diverse and may be divided into three categories: straws, flowers, and plants. The carbonization time and temperature ranges remain vast, ranging from 1 to 3 hours and 600 to 900 degrees Celsius, respectively. Carbon from biomasses such as camellia (W7) or reed flowers (W11), as well as catkins of various varieties (W8 – W10), has been examined in Li–S cells in the floral category.
Agricultural Food Biomass
According to the research, the three most common activating reagents utilized in this group were KOH, ZnCl2, and H3PO4, with KOH dominating. In terms of calcination temperature, a wide range is seen, ranging from 550 to 1000 C. The publication by Li et. al. in 2019 added to the body of knowledge for researchers interested in using corncob waste (and other biomass leftovers) as a sulfur host.
Aquatic Biomass
According to the research, owing to the low cost, high carbon content, and notably rapid development, some marine aquatic plants are ideal and sustainable candidates for manufacturing porous (C) or activated carbons (AC). The SBET and V p values are higher than those of the non-activated ones, as are their S content of 80 %.
Animal and Human Waste Biomass
According to the most recent research, the use of KOH as an activating agent for this biomass type remains dominant. When compared to H3PO4, this method yields greater SBET and V p values while also requiring lower carbonization temperatures.
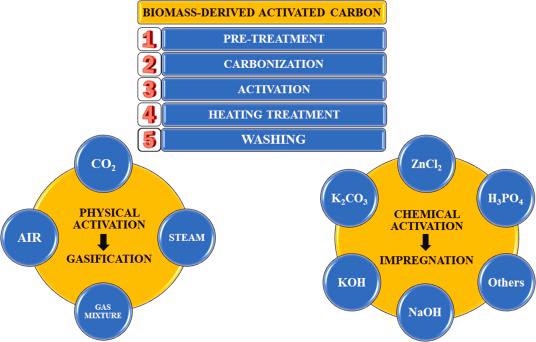
General preparation procedure and activation methods of biomass-derived activated carbons. Image Credit: Benítez, A et al., Renewable and Sustainable Energy Reviews
Household and Industrial Waste
Only KOH was utilized as an active catalyst for this set of carbons, and the carbonization duration and temperature ranges were also varied. Activation increases SBET and V p values, as predicted, however the composite with the greatest S content (70%) is found in a non-activated carbon with the lowest Vp value.
The latest research focuses on a major point that although various biomass types are present, the most fundamental constituent is carbon. Carbons have a multilayer pore structure, high SSA, high porosity, and a high concentration of heteroatoms that persist after carbonization or being injected during activation, and appear to be preferred in general.
Although the research has mentioned core aspects, several factors such as additional costs due to AC generation, quantitative evaluation of the volume of biomass required, and industrial expansion because of future requirements are still yet to be carefully assessed. Similarly, the problems related to agri-food biomass utilization such as large land requirements and the presence of impurities are still to be addressed.
In short, the low cost and especially very little harmful environmental emissions are the core advantages that have motivated the utilization of biomass in power generation. Such a variety of biomass truly makes it the best possible choice to be used in the prevailing lithium-sulfur batteries.
References
Benítez, A., Gahete, J. A., Chien, Y. C., Caballero, A., Morales, J., & Brandell, D. (2021). Recent advances in lithium-sulfur batteries using biomass-derived carbons as sulfur host. Renewable and Sustainable Energy Reviews. https://www.sciencedirect.com/science/article/pii/S1364032121010522
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.