Keratins, like many other naturally generated macromolecules, have bioactivity and biocompatibility built-in. As a result, rheological measurements were used to maximize an innovative injectable KRT-FIB hydrogel, as well as its injection achievement, swelling behavior patterns, and morphologies.
Keratin-Conjugated Fibrinogen (KRT-FIB) Composition
Researchers found that the novel KRT-FIB-injectable hydrogel has outstanding viability, proliferation, and migration/cell–cell engagement, implying that it is a favorable choice for oral tissue repair with high clinical potential application. Traumatic injury to the oral cavity is unusual, and it is frequently accompanied by uncontrollable bleeding and inflammatory responses.
Because of their simple composition, minimally invasive strategy, and site-specific distribution, injectable hydrogels have indeed been considered as promising candidates for the treatment of oral injury issues. Because of their uniqueness, fibrinogen-based hydrogels were extensively researched as effective substances for the healing process in tissue engineering.
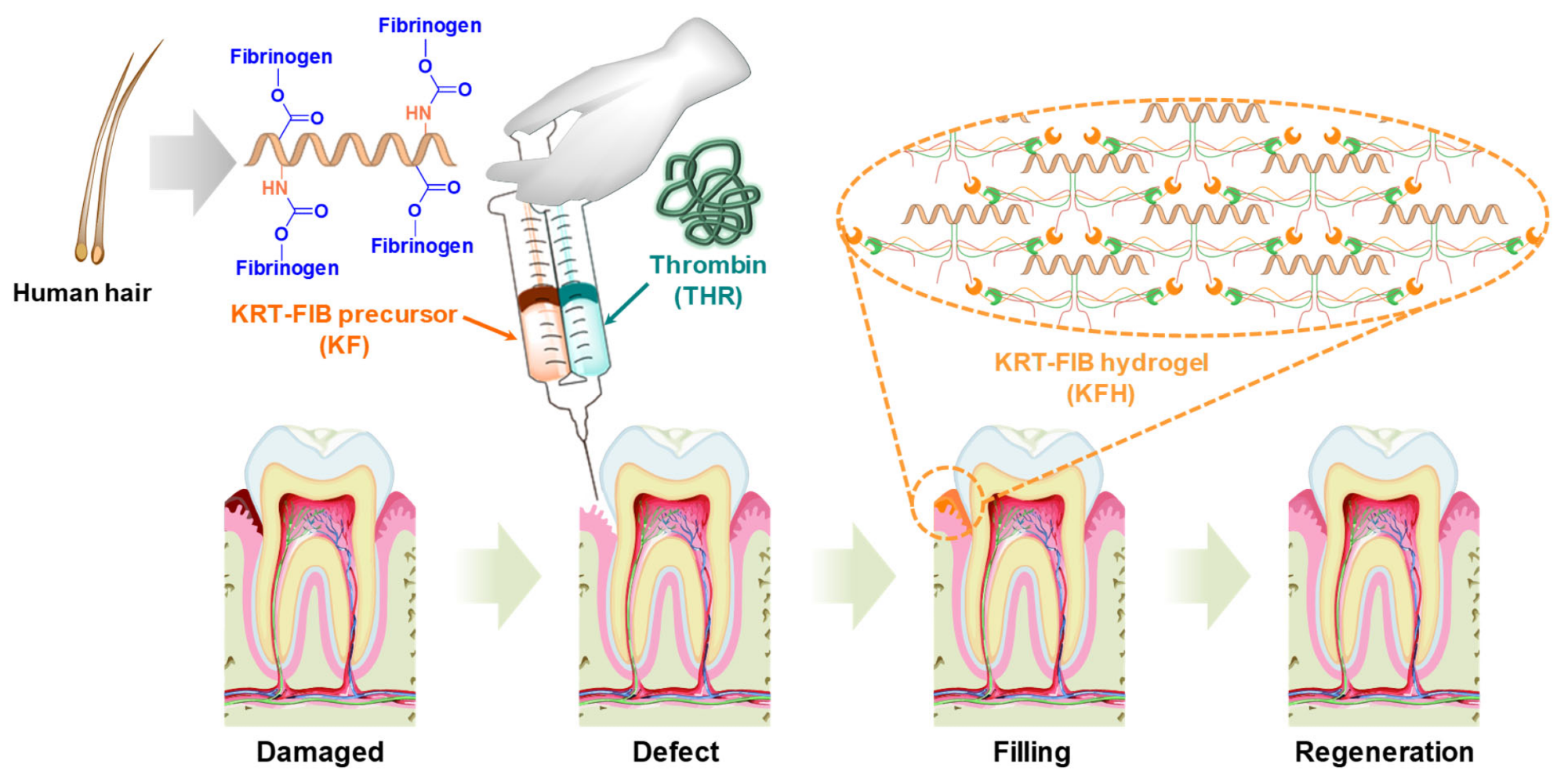
Schematic illustration of keratin-conjugated fibrinogen (KRT-FIB) hydrogels (KFHs) for oral tissue regeneration. Image Credit: Kang, H., et al., International Journal Molecular Science
Keratin (KRT) is a non-soluble type of protein found in epidermal appendageal formations, hair, cuticles, horn, gallops, wool, and feathers. KRT plays critical structural and protective roles, especially in the epithelium. Along with its intrinsic bioactivity, bio-compatibility, and physicochemical characteristics, human hair-derived KRT is highly appealing for wound healing therapy, as researchers recently reported.
Amidst these benefits, previous KRT-based hydrogel research has been limited to KRT proteins derived from wool or feathers. Furthermore, hydrogels based on KRTs derived from human hair, particularly injectable systems, have not been studied for oral tissue repair.
Fibrinogen as Wound Healing Process
Fibrinogen (FIB) is a promising candidate because of its inherent wound healing process, which enhances reparative cellular activities, as well as its angiogenic and anti-inflammatory properties, both of which are required for wound healing and tissue repair.
FIB is a protein that is important in regulating angiogenesis and bone regeneration. The creation of FIB polymeric materials between transglutaminase factor XIII and calcium ions in the existence of thrombin results in the very first clinically approved FIB hydrogel for tissue repair (THR).
Moreover, due to the limited design ability of a protein molecule, current FIB hydrogels have low mechanical resilience, porosity, and adhesive characteristics. Hydrogels containing only FIB are supposed to improve scaffold contraction and thus do not support continuous tissue growth. As a result, when transplanted, such FIB hydrogels are relatively stiff and hard to handle.
To address these issues, the authors created an injectable KRT-FIB hydrogel (KFH) for oral tissue repair. Human hair-driven KRT proteins have been covalently linked with FIB enzymes via a simple coupling reaction to accomplish homogeneous KFH formation.
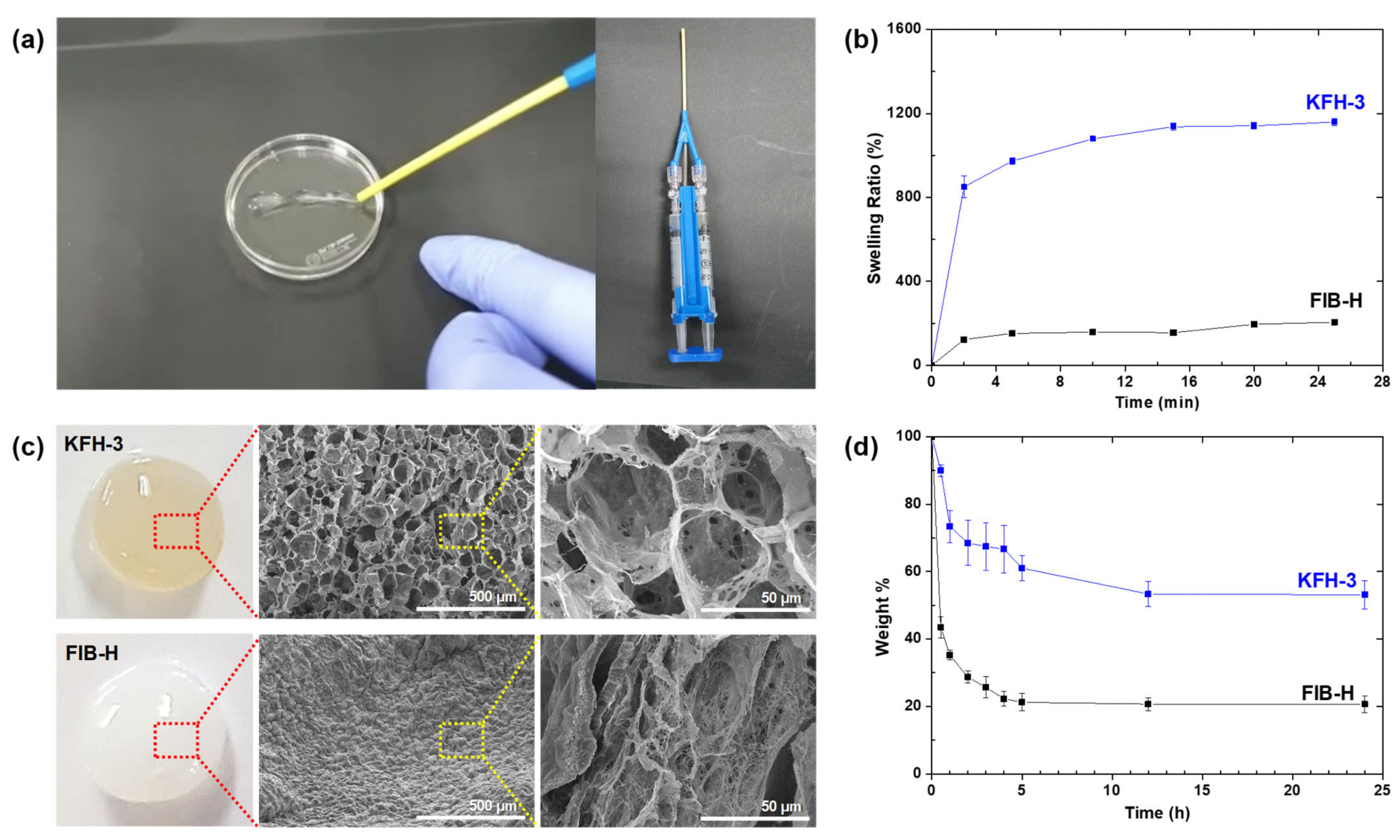
(a) The KFH-3 hydrogel was able to pass through a needle without clogging, indicating its high injectability (left); the precursor solution was loaded onto one side of a dual-chambered syringe with an 18 G needle (diameter = 1.3 mm), and the THR solution was loaded onto the other side (right). (b) Swelling kinetics of KFH-3 and FIB-H in deionized water at 37 °C. (c) Digital pictures and SEM images of KFH-3 and FIB-H at different magnifications (scale bar = 500 μm and 50 μm, respectively). (d) Temperature- and pH-responsive degradation study of hydrogels was evaluated by the dry weight ratio of the hydrogel samples. Image Credit: Kang, H., et al., International Journal Molecular Science
Method of Making KRT-FIB
Despite the fact that both KRT and FIB are hydrophilic peptides, they cannot be combined into relatively homogenous hydrogels when THR is present. To address this issue, covalently linked KRT-FIB components were created using a simple coupling reaction. In addition, the effect of KRT conjugation was determined by evaluating its thermal performance using thermogravimetric analysis (TGA).
Hydrogels must be crafted with several crucial physicochemical parameters in mind in order to accomplish clinically acceptable injectability shear-thinning activity and enough strength to withstand permanent deformation.
Complex viscosity is an important variable to consider when designing injectable systems because it quantifies the hydrogels' ability to react to adjustments in shear stress during injection as well as resist deformation inside a tissue after injection. Extrusion research was carried out to investigate the injectable effectiveness of the KFH. Injectable hydrogels were created using a dual-chambered syringe.
Result of the Research
One-way statistical significance was used for data analysis of Tukey's retrospective analysis for relevance. The intravenous KFH was developed and tested to improve porosity and viscosity by monitoring the molar ratio of KRT and FIB.
The generated KFH demonstrated synergistically enhanced proliferation of encapsulated HGFs, and HGFs displayed diffuse morphological characteristics when encapsulated in KFH. These findings indicate that the novel KRT-based hydrogel has a significant potential for use as a scaffold for tissue regeneration in medical applications.
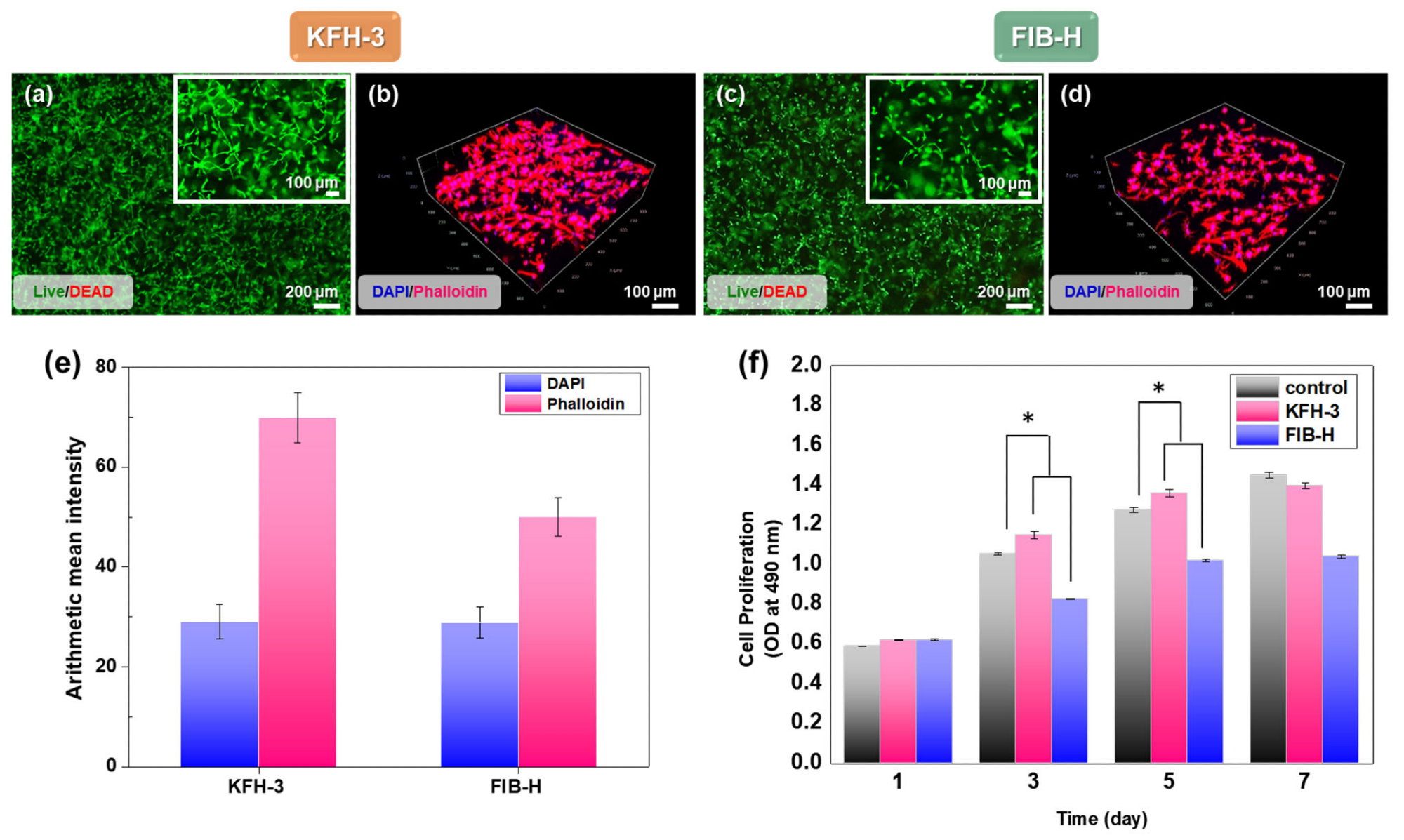
Characterization of cell adhesion and proliferation in three-dimensional cell encapsulation in FIB-H and KFH-3. For the cell viability assay, human gingiva fibroblasts (HGFs) embedded in FIB-H and KFH-3 were stained with calce-in_Am (green)/ethidium homodimer (red). LIVE/DEAD assay 24 h after encapsulation (a,c) shown at low (scale bar = 200 µm) and high (scale bar = 100 µm) magnification. (b,d) Confocal pictures of encapsulated HGF cells labeled with 2-(4-aminophenyl)-1H-indole-6-carboxamidine (DAPI) and Phalloidin (F-actin) (scale bar = 100 µm). (e) Expression of DAPI and phalloidin was quantified by immunofluorescence after 3 days in culture. (f) The proliferation of HGF cells encapsulated in KFH-3 and FIB-H for 7 days, as measured in a CCK-8 assay. Both data were evaluated for cyto-compatibility at a concentration of 20 mg/mL. Data are presented as the mean ± SD of triplicate experiments: * p < 0.05. Image Credit: Kang, H., et al., International Journal Molecular Science
Usage of Keratin-Conjugated Fibrinogen
Tissue engineering has suggested the advancement of materials science for implantable porous scaffolds at the wounded area, which acts as temporary supporting materials for cell ingrowths and degrades alongside extracellular matrix producing vascularization, and tissue regeneration.
Keratin is a fibrillar protein that is found in the extracellular matrix that provides an outer covering for wool, hair, feathers, and nails, as well as a substitute for collagen biopolymers. The high cysteine substance of keratin aids in the immobilization of biologically active compounds for drug delivery applications.
References
Kang, H., et al. (2021). Injectable Human Hair Keratin–Fibrinogen Hydrogels for Engineering 3D Microenvironments to Accelerate Oral Tissue Regeneration. International Journal Molecular Science 2021, 22(24), 13269; https://www.mdpi.com/1422-0067/22/24/13269
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.