Photocatalysis provides a long-term solution for converting solar energy into high-value chemical fuels. Over the last few decades, a lot of effort has gone into discovering and designing better photocatalysts. Despite this, they have poor light absorption and responsiveness, simple recombination of photogenerated carriers, and a slow surface catalytic reaction rate.
To improve the performance of photocatalysts, a variety of solutions have been proposed including doping heteroatoms, adding cocatalysts, and creating heterojunctions. These enhancements frequently make catalyst systems more difficult and expensive. The absence of efficient photocatalysts is still a barrier to their widespread use.
Because of their decreased carrier migration distance and high surface area, two-dimensional (2D) materials have enhanced photocatalytic activity and a substantially higher density of catalytically active sites than their bulk counterparts. Because of their unique physical/chemical properties, rare-earth minerals have been extensively studied as catalysts for a variety of chemical reactions. Rare-earth elements, in particular, are frequently utilized as dopants to improve the photocatalytic activity of the 2D semiconductors. Only a few experiments using 2D rare-earth materials are documented so far.
High-throughput computations based on the density functional theory (DFT) calculations are effectively used to speed up material discovery in a variety of applications. Various hypothetical 2D materials have been described in the literature since the emergence of large-scale computational materials databases. Despite this, only a small number of theoretically forecasted 2D materials have been successfully synthesized in trials.
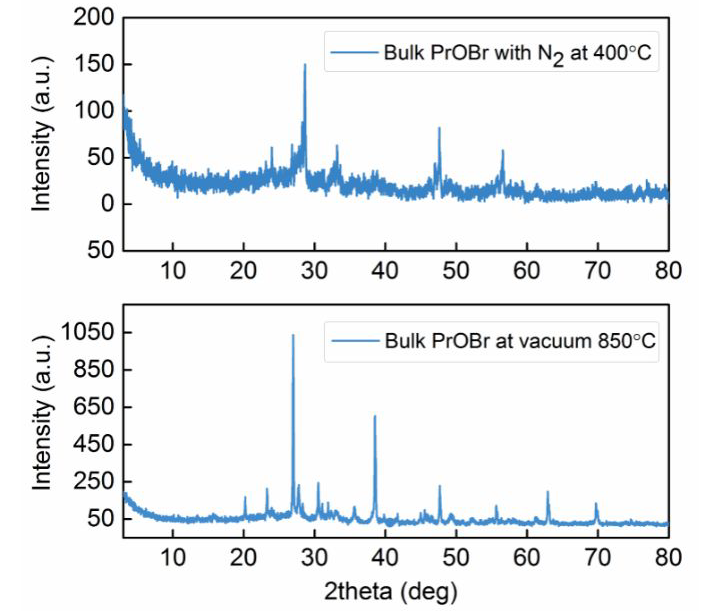
XRD patterns of bulk PrOBr treated with N2 at 400 °C for 8 h and treated under vacuum at 850 °C for 12 h. Image Credit: Noori, A. W et al., ACS Energy Letters
About the Study
In this study, the authors discussed the discovery and production of praseodymium oxyhide (PrOBr), a two-dimensional (2D) rare-earth material, as an efficient photocatalyst. The potential of 2D PrOBr as a promising photocatalyst with strong stability and activity was assessed by doing high-throughput screening over popular computational 2D materials databases. Exfoliation of bulk PrOBr powders was used to successfully synthesize 2D PrOBr. The proposed 2D material's intrinsic photocatalytic capabilities were also confirmed.
The team used a high-throughput screening method to identify a 2D rare-earth material, PrOBr, for photocatalysis based on DFT-calculated structural and electrical characteristics. An ultrasonic exfoliation process was used to successfully produce this newly found 2D material from its bulk precursor. Various characterization techniques were used to identify PrOBr's 2D structure. The hydrogen evolution process (HER) and carbon dioxide reduction reaction (CO2RR) were efficiently catalyzed by 2D PrOBr, to demonstrate its photocatalytic capability. The ability of 2D PrOBr to balance inherent adsorption and charge separation efficiency was determined to explain its outstanding photocatalytic performance.
The researchers utilized phase stability and exfoliation energy to assess the synthesizability of the proposed 2D material. The energy over a linear combination of the stable phases in the 0 K DFT phase diagram, commonly known as Ehull, was calculated to determine the phase stability of 2D materials.

(a-c) SEM images of 2D PrOBr. EDX mapping images of Pr, Br, and O (df) in c. Image Credit: Noori, A. W et al., ACS Energy Letters
Observations
The yields of CO and CH4 on 2D PrOBr were significantly higher than on 2D C3N4 and BiOCl. The evaluated CH4 and CO and generation rates of 0.862 μmol.h-1.g-1, and 17.17 μmol.h-1.g-1, respectively, were several times higher than those of usual photocatalysts. With the Tauc-plot empirical formula, the empirically measured band-gap of the 2D PrOBr was 2.19 eV.
The band gaps of La2GeI2 and NdOBr were 1.29 and 1.43 eV, respectively, and were well-positioned in the near-infrared region. The band gaps for HoTe2ClO5 and PrOBr determined by mTASK were 2.37 and 2.18 eV, respectively. Ehull was less than 30 meV/atom in all of the proposed 2D materials, which was a conservative phase stability threshold employed in the literature.
The measured hydrogen evolution rate of water splitting and methane production rate of carbon dioxide reduction was 60.28 μmol.h-1.g-1 and 0.862 mol.h-1.g-1, respectively, which were about 3-6 times and 7 times greater than conventional 2D photocatalysts such as 2D C3N4 and BiOCl. The production rate of H2 and the conversion rate of CO2 were found to be at least twice that of their bulk counterparts, and much greater than that of ordinary non-modified 2D photocatalysts such as 2D C3N4 and BiOCl.
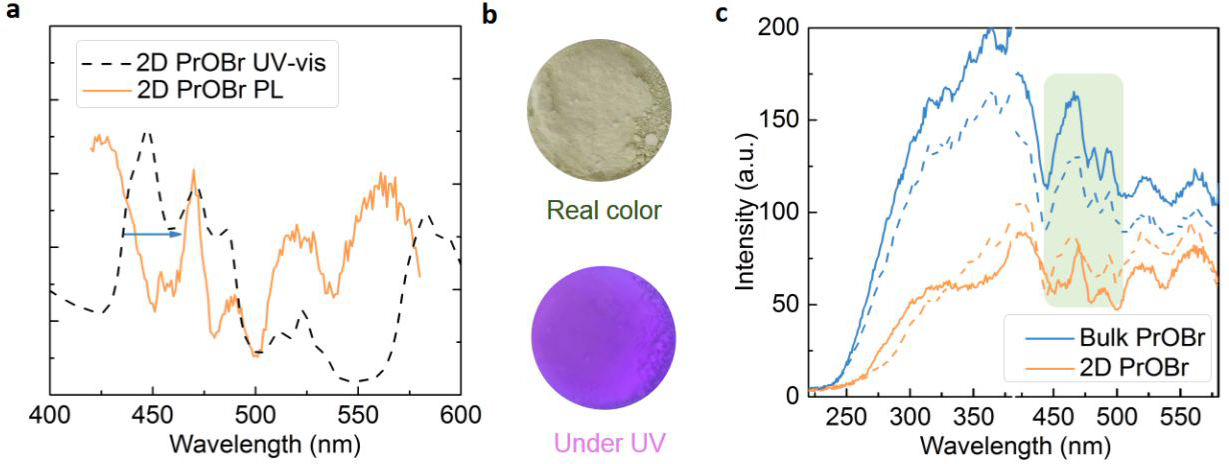
(a) UV-Vis absorption spectrum and PL spectrum of 2D PrOBr. (b) Real color images of 2D PrOBr powder and the powder under 365 nm irradiation are shown. (c) PL spectra obtained under the excitation wavelength at 220 nm of bulk and 2D PrOBr from batch to batch. Image Credit: Noori, A. W et al., ACS Energy Letters
Conclusions
In conclusion, this study elucidated the development of a 2D rare-earth material, PrOBr, as a photocatalyst for HER and CO2RR using a combination of computational high-throughput prediction and experimental validation. High-throughput computations with a tiered property of photocatalysis interest first indicated 2D PrOBr as a potential option. The bulk material was then treated with ultrasonic exfoliation to prepare it.
The authors observed that for HER and CO2RR, 2D PrOBr has good photocatalytic activity. They believe that with some adjustments, this new 2D photocatalyst could be used in additional photocatalytic fields.
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.