Medical tissue adhesives are effective wound closure methods since they cause less pain and fewer secondary wounds in comparison to conventional staplers or sutures. An optimal bioadhesive has several properties such as (1) in vivo biodegradability and biocompatibility, (2) adhesion flexibility, (3) minimal hypersensitivity or toxicity, (4) adequate preparation time, (5) sufficient adhesive strength, (6) storage stability, and (7) low swelling index and ease of application.
Normal tissue function and structure can be achieved by the use of surgical binding of the damaged tissues that would withstand the tensile stress that the tissues are subjected to while simultaneously preventing body fluid leakage. Conventional methods such as staplers, external ears, and sutures have multiple disadvantages such as trauma to the surrounding tissues, prevention of natural flow of body fluids, air, and blood, along with poor accuracy on the intended tissues that are difficult to access.
SF proteins from spider silk can be effective in increasing the hemostatic effect by holding onto the nearby bleeding site, along with enhancing wound regeneration and bio-adhesion function, biodegradability, and biocompatibility while being biohazard free. In this study, the team used click chemistry reactions to synthesize bioadhesives from non-swellable SF derived from spider silk with the use of tannic acid as a bioadhesive agent. Additionally, the team attempted to produce a bone-specific bioadhesive using hydroxyapatite (HAP) since HAP efficiently adsorbs onto proteins.
A schematic summary of two bioadhesives. Image Credit: Lee, W.-J et al., Materials
Materials and Methods
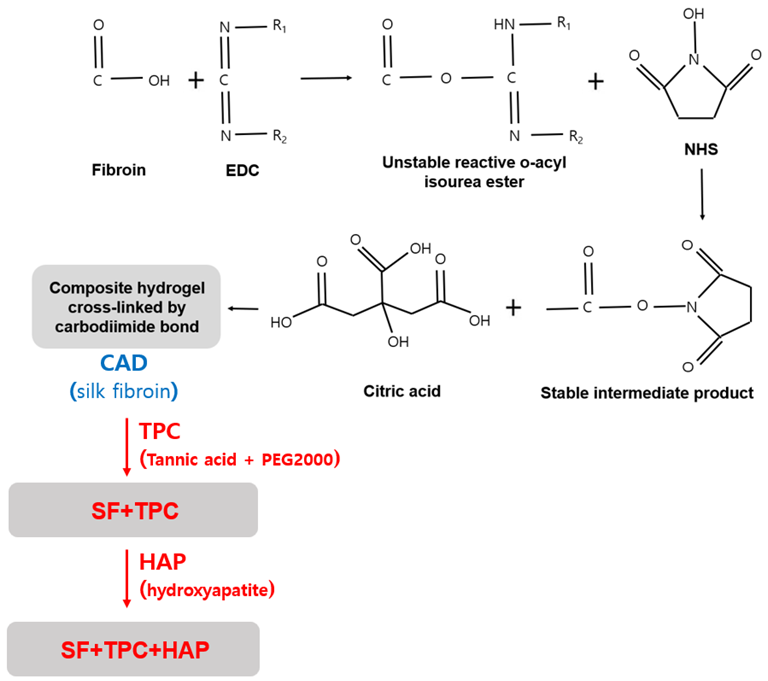
In the present study, the researchers used included citric acid, tannic acid, N-Hydroxysuccinimide (NHS), and 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC), and chondroitin-6-sulfate. The bioadhesive was derived from spider silk with polyethylene glycol with a molecular weight of 2000 (PEG2000). Two types of adhesives were produced. One was a citric acid derivative (CAD), which is a precursor of SF protein with tannic acid and PEG2000 curing agents (TPC) (SF + TPC), and the other was SF + TPC with the addition of HAP (SF + TPC + HAP).
The SF protein components were subjected to component analysis through gas chromatography/mass spectroscopy (GC-MS). This was followed by the determination of the contained amino acid through an amino acid analyzer. Furthermore, chondroitin-6-sulfate content was estimated using a chondroitinase waters’ 515 pump and a 486 UV detector, while the chemical structure of the SF protein extract was analyzed by infrared spectroscopy.
The hydrophobic and hydrophilic properties of the SF protein were determined by measuring the contact angle. Additionally, a universal testing machine (UTM) equipment measured the adhesive strength of the SF protein extract on pig skin. The cytotoxicity of the SF protein bioadhesive was determined as per ISO 10993-5. Finally, a test-tube tilting method was used to test the gelation time, while the consequences of the bioadhesive on osteogenic differentiation were investigated using alkaline phosphatase (ALP).
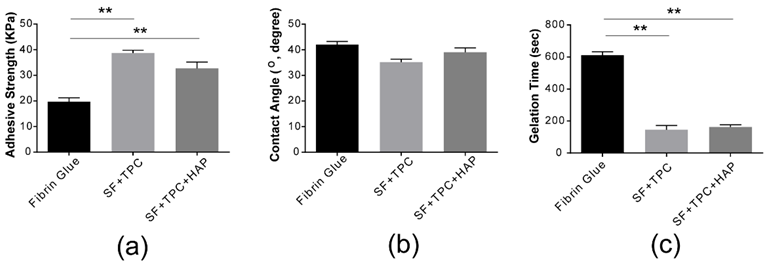
Structural analysis. (a) The adhesive strength of conventional fibrin glue was compared to silk fibroin protein in skin (SF + TPC) and in bone (SF + TPC + HAP). Silk fibroin showed about two times higher adhesive strength than conventional fibrin glue. (b) The contact angle of conventional fibrin glue was compared to silk fibroin protein in skin (SF + TPC) and in bone (SF + TPC + HAP). Overall, SF showed a lower contact angle compared to fibrin, suggesting greater hydrophilicity. (c) The gelation time. SF, silk fibroin; TPC, tannic acid and polyethylene glycol 2000 as curing agents; HAP, hydroxyapatite. n = 5. ** p < 0.001. Image Credit: Lee, W.-J et al., Materials
Observations
The GC-MS peaks exhibited more than 85% glycyl-L-proline (C7H12N2O3) content in the SF samples. The synthesized SF bioadhesive showed about two times greater adhesive strength as compared to conventional fibrin glue. The hydrophilicity of SF + TPC + HAP samples was greater than that of fibrin glue with a contact angle of 38 ±7°. The greater the hydrophilic properties, the higher the cohesive force with which the bioadhesive attaches to the skin. In comparison to fibrin glue, SF exhibited a gelation time of less than three minutes.
Furthermore, the cytotoxicity evaluation of the bioadhesives showed a cell viability of 86±12.23% at 10 mg/L, higher than that of the fibrin adhesives at 78.2±10% at 10 mg/L. The increased ALP activity observed in the SF + TPC + HAP groups suggested enhanced initial cell survival and differentiation.
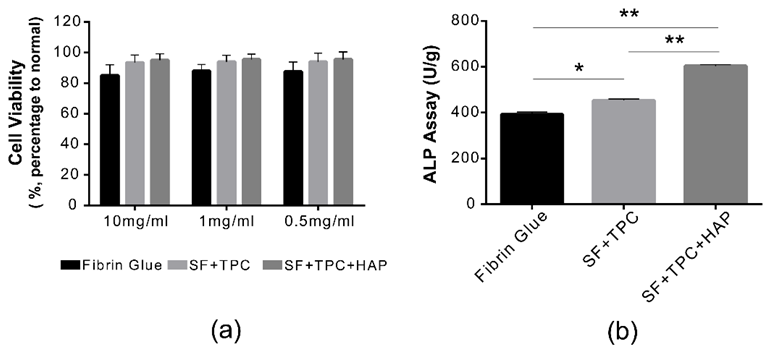
Functional analysis. (a) Cell viabilities were measured in both undiluted (10 mg/mL) and diluted states (1 mg/mL and 0.5 mg/mL) of both fibrin and silk fibroin proteins. There was no difference in cell viability among conventional fibrin glue, silk fibroin protein in skin (SF + TPC), or silk fibroin protein in bone (SF + TPC + HAP). There was no cytotoxicity at 10 mg/mL. (b) ALP activity was presented as U/g in each group of fibrin, SF + TPC, and SF + TPC + HAP. SF, silk fibroin; TPC, tannic acid, and polyethylene glycol 2000 as curing agents; HAP, hydroxyapatite. n = 5, * p < 0.05; ** p < 0.001. Image Credit: Lee, W.-J et al., Materials
Conclusions
To summarize, the team produced bioadhesives from SF protein made of spider silk to assess its compatibility as tissue adhesives used in wound closure. The strength tests using pig skin showed two times stronger adhesion for SF compared to fibrin glue. Additionally, SF bioadhesives can be injected with lower gelation times and minimum cell cytotoxicity, and combined with stronger adhesion can accelerate wound healing by effectively reducing and closing the tissue gap.
Finally, the introduction of collagen can increase adhesion strength through the formation of a dual network with SF bioadhesives. According to the authors, SF bioadhesives are a promising medical tool for the efficient and effective closing of surgical wounds, mainly bone fractures.
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.
Source:
Lee, W.-J.; Cho, K.; Kim, A.-Y.; Kim, G.-W. Injectable Click Fibroin Bioadhesive Derived from Spider Silk for Accelerating Wound Closure and Healing Bone Fracture. Materials 2022, 15, 5269. https://doi.org/10.3390/ma15155269