Significant studies have been carried out over the last decade on synthesizing biopolymers using renewable resources to substitute environmentally hazardous synthetic polymers. Polylactic acid (PLA) is biodegradable, versatile, and derived entirely using natural resources. Although PLA has several advantages when it comes to performance and production, its poor mechanical characteristics, such as low heat resistance and brittleness, limit its applicability.
Several PLA composites featuring various fillers have been devised in the past decade to address these shortcomings and generate environmentally beneficial polymers with excellent physiochemical characteristics. GFs are appealing because of their optimal heat resistance, mechanical properties, relatively low cost, and simple production. several GF characteristics may be changed during the fabrication of PLA-glass composites, and a detailed understanding of the structural properties and composition is required to fully realize their potential.
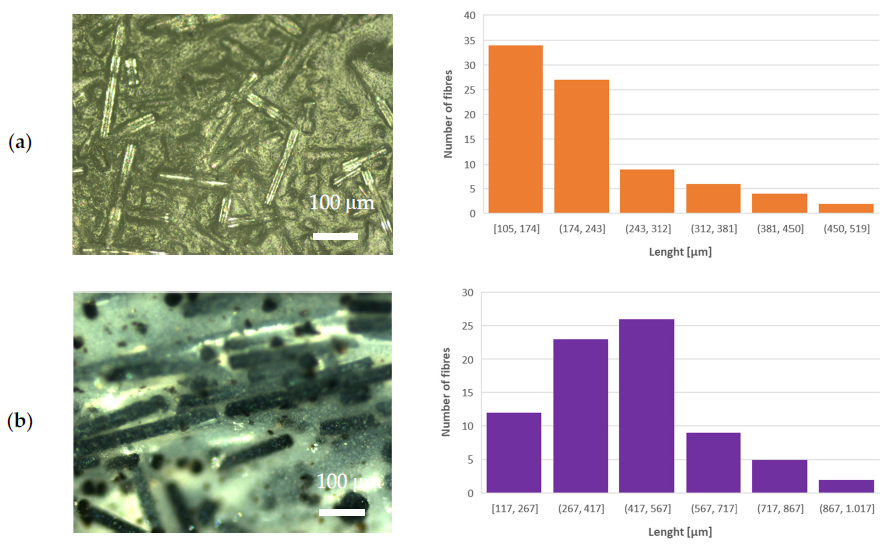
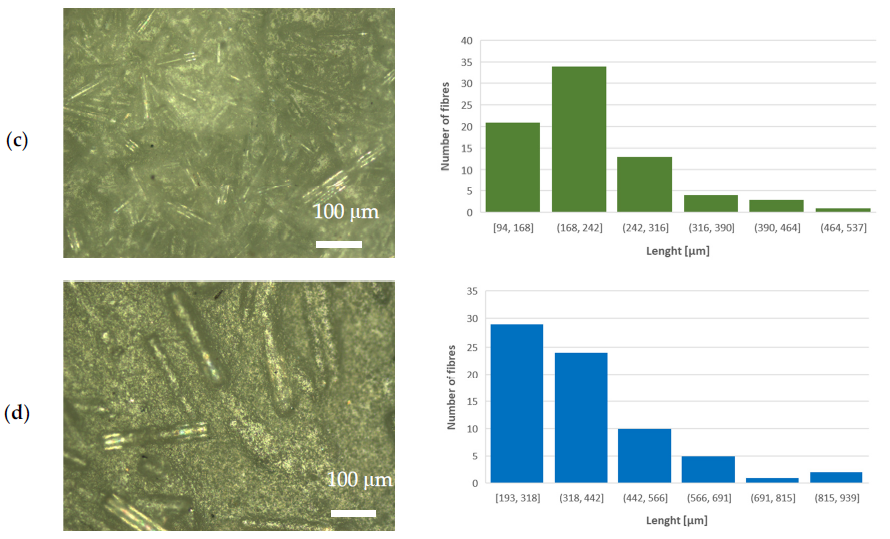
Optical microscope images (left-hand side) and length distribution of milled fibers (righthand side) of (a) PLA–WG, (b) PLA–IPG, (c) PLA–HYBG, and (d) PLA–ARG composites. Image Credit: Klaser, T et al., Polymers
About the Study
In this study, the team employed four kinds of GFs to synthesize glass composites, including iron phosphate glass fibers (IPG). In the laboratory, GFs with the composition 40Fe2O3-60P2O5 in mol% (IPG) were created. PLA-glass composites were prepared through PLA dissolution in an organic solvent and GF solution.
A diffractometer was used to obtain X-ray diffraction (XRD) data at room temperature. The Eva software was used to identify the PLA and composite constituents. Thermal analysis was conducted at temperatures ranging from 20 to 200 °C at a 10 K/min heating rate.
A polarizing microscope was used to examine the cross-section and surface morphology of the PLA-glass composites, and impedance spectroscopy (IS) was used to evaluate the electrical characteristics of the synthesized composites. To confirm if different glass types could improve the PLA crystallization process, heating of the PLA-glass composites for 10 minutes at 210 °C was carried out, followed by annealing for an hour at 90 °C. The annealed composites were examined using XRD following heat treatment.
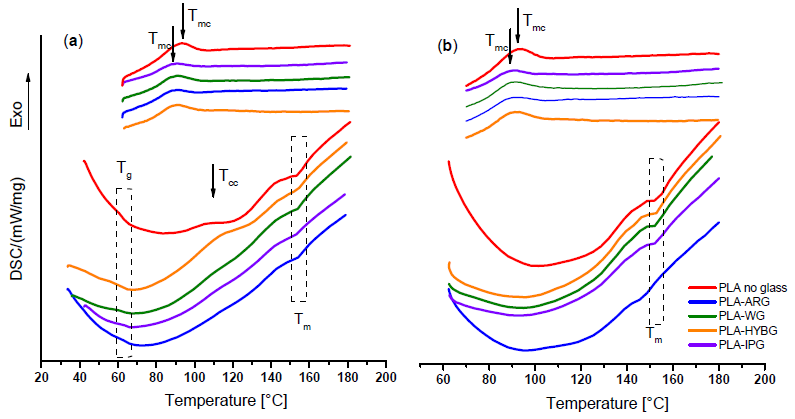
DSC curves for the neat PLA and PLA–glass composites in (a) first and (b) second heatingcooling cycle. Image Credit: Klaser, T et al., Polymers
Observations
The industrial grade PLA's XRD diffractogram exhibited strong diffraction lines attributable to talc and calcite impurities overlaid on a large halo associated with amorphous PLA. Moreover, the diffractograms of the synthesized PLA-glass composites were similar to those of the PLA, exhibiting that neither the GFs nor the PLA were crystallized. The PLA-glass composites with heat treatment did not exhibit any new diffraction lines in their XRD patterns, indicating that the PLA crystallization was not initiated with any kind of GF and that all composites continued to remain amorphous.
The lack of crystallization following heat treatment could be attributed to the loose bond between the PLA matrix and the GFs. As a result, if a substantial amount of interaction between PLA and the GFs was achieved, it could be concluded that the fibers can induce crystallization of the PLA matrix after heat treatment. Furthermore, the scanning electron microscope (SEM) imagery revealed that the GFs in all composites were randomly distributed and oriented within the PLA matrix.
The cross-section images showed that all GF types form a weak bond with the PLA matrix. In this study, the surface of the GF was not altered to enhance the polymer bond interaction. As a result, a slightly weaker interaction between the PLA matrix and the GFs was expected in these composites.
In the initial heating run, the DSC curves for the PLA-glass composites, regardless of the GF types, displayed extremely comparable characteristics to those of the plain PLA, demonstrating that the inclusion of GFs did not affect the melting temperature or glass transition. This signified that all GFs could inhibit the PLA crystallization, possibly due to weaker GF and PLA matrix interactions for nucleation sites and/or excessive GF amounts, which prevent the development of large crystalline domains.
Additionally, DC conductivity measurements indicated that all the PLA-glass fiber composites exhibited Arrhenius behavior; that is, linear temperature dependency throughout the whole temperature range. These results implied substantial high-temperature stability of the PLA-glass fiber composites compared to neat PLA.
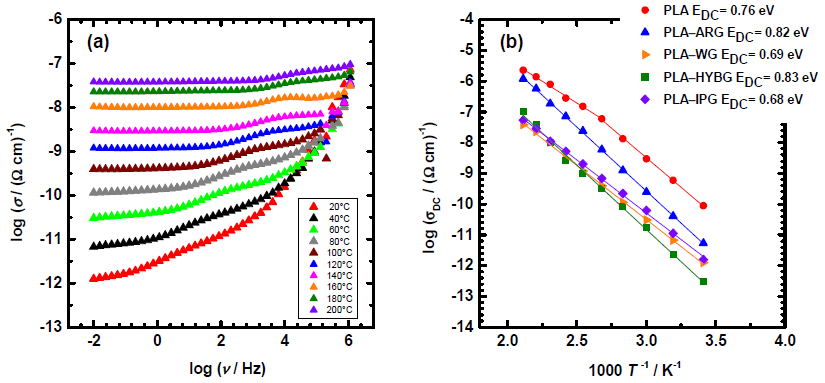
(a) Conductivity spectra at different temperatures for PLA–WG composite and (b) Arrhenius plot of DC conductivity for all composites. Image Credit: Klaser, T et al., Polymers
Conclusions
To summarize, the researchers demonstrated the synthesis of PLA-glass composites with technical grade PLA and different GF types in a weight ratio of 1:1. Compared to neat PLA, these composites exhibited superior thermal stability, mechanical strength, and insulating qualities.
As implied by the Arrhenius behavior, the inclusion of all kinds of GFs enhances the insulating qualities by lowering conductivity while also broadening the thermal stability range of the composites. According to the authors, these composites may be ideal for small-scale disposable usage in indoor and outdoor construction applications where the material's in-situ degradation is required.
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.
Source:
Klaser, T.; Balen, L.; Skoko, Ž.; Pavić, L.; Šantić, A. Polylactic Acid–Glass Fiber Composites: Structural, Thermal, and Electrical Properties. Polymers 2022, 14, 4012. https://www.mdpi.com/2073-4360/14/19/4012