Single-atom catalysis has the potential to be a revolutionary technology in chemistry. Since its inception in 2011, there has been significant progress in research into this technology. Several studies have investigated factors such as the reaction environment’s influence on single-atom catalysis and deactivation pathways.
Single-atom catalysis combines the benefits of highly selective homogenous catalysts employing organometallic complexes and heterogenous catalysts, where catalyst separation is not required and offers the advantage of stable performance over prolonged periods of use.
Recent research (including by the authors of the new paper) has indicated the viability of single-atom catalysis for applications such as water electrolysis, hydrogen fuel cells, and chemical manufacturing. The industrial and commercial uptake of single-atom catalysis will depend on the development of single-atom catalysts with sufficient metallic load and sufficient stability over prolonged periods of time.
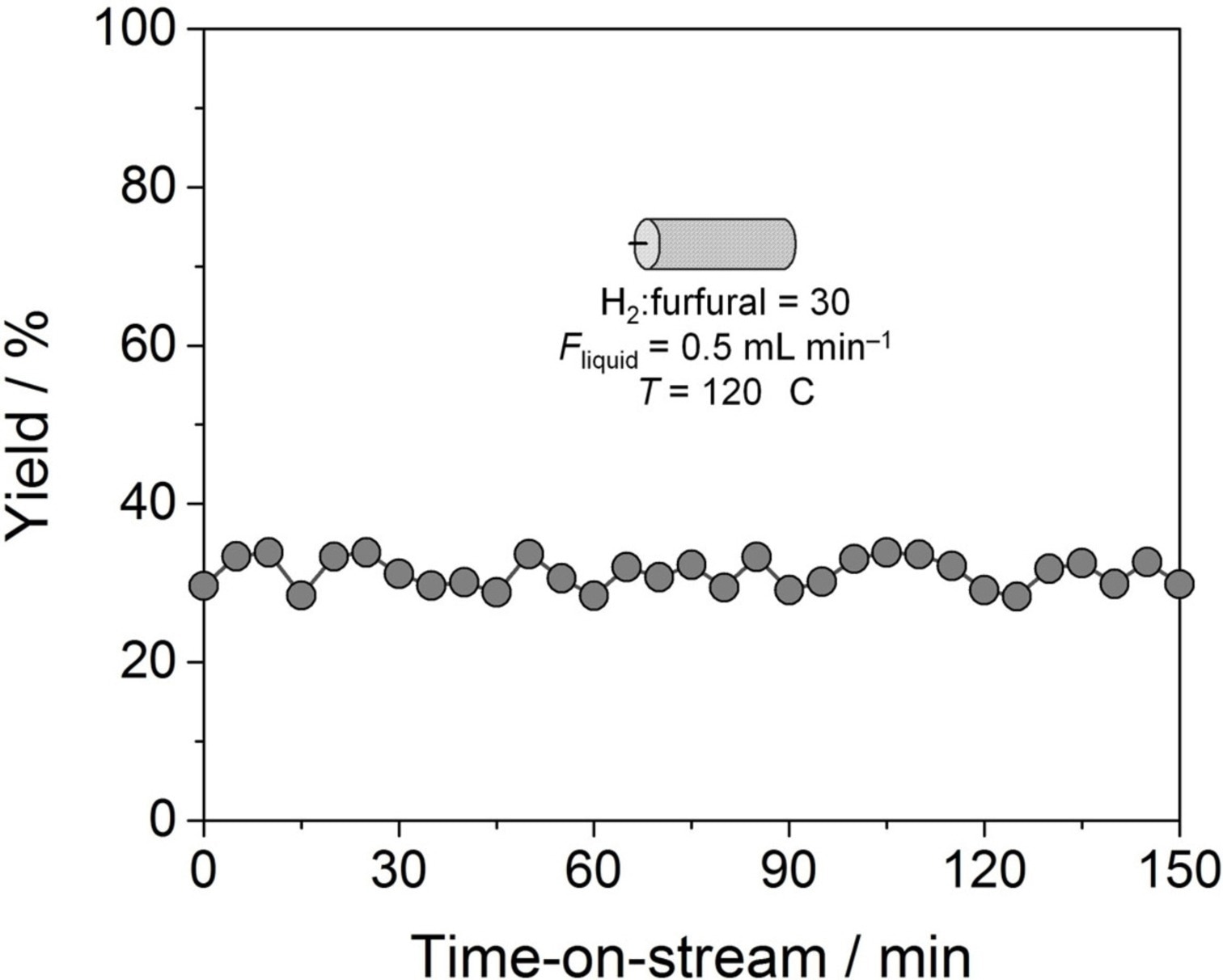
Hydrogenation of furfural over CSMs coated with Cu1@mpgC3N4. Image Credit: Pagliaro, M et al., ChemCatChem
Advances in the development of leach-poof metal catalysts with high selectivity and progress in flow chemistry are helping to revolutionize the chemical industry and can help to overcome the historical reluctance of the fine chemical industry to embrace metal transition catalysts.
Technical perspectives are not the only consideration that the chemistry industry must make, however. The financial viability of single-atom catalysis must also be demonstrated sufficiently if this potentially game-changing technology is to achieve widespread acceptance and uptake in the fine chemical industry and associated sectors.
The industry is currently undergoing a profound shift to smaller, decentralized chemical production using advanced catalysis technologies under continuous flow conditions. This shift will allow the production of high-value and low-volume chemical products with minimal waste generation.
The Paper
The new paper in ChemCatChem aims to close the techno-economic cycle in studies on heterogeneous catalysts and uncover the viability of single-atom catalysis as an organic process technology in the chemical industry.
Studies over the past decade have highlighted the potential of single-atom catalysis to transition from a fundamental research topic to one of practical relevance in the chemical industry. Utilizing metal complexes, organocatalysts, heterogenized metal nanoparticles, or enzymes in heterogenous catalysis approaches can fulfill the objective of lean goods manufacturing in industrial-scale production plants.
The authors have stated that a technical advantage of single-atom catalysis for chemical production is its potential employment in catalytic microreactors, which is highly synergistic in nature.
Several methods have been developed in recent studies for synthesizing catalytic material for single-atom catalysis which possess high metal loads, including precursor dilution strategies and pyrolysis-deposition methods. Catalysts have been synthesized with large surface areas, pore-specific volumes, and, importantly, high metal load. Research has confirmed the activity and stability of several suitable catalysts.
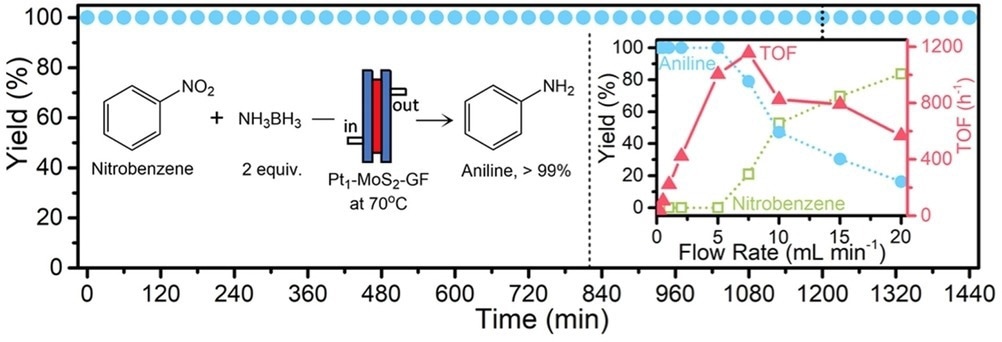
24 h on-stream demonstration of the Pt1-MoS2-GF catalyst in the quantitative conversion regime of nitrobenzene to aniline under flow. Condition: 0.025 M nitrobenzene with 0.050 M ammonia borane in acetonitrile/H2O mixture (5 : 1, v/v) at a flow rate of 1 mL/min. Yield vs. flow rate test shown in the inset as performed at 20 h. Image Credit: Pagliaro, M et al., ChemCatChem
Research has revealed that single-atom catalysts employed under flow conditions display the same stability as those under batch conditions. This proof of concept is incredibly important for the industrial uptake of this method for chemical production. Two major methods have been developed for the large-scale production of suitable single-atom catalysts, which opens the route toward employing several potential leach-proof single-atom catalysts under flow.
One of the main advantages of this catalysis approach is its suitability for organic chemistry reactions that require trace amounts of catalyst. Another challenge is the contamination of trace metal catalysts by impurities during reactions, and the use of single-atom catalysis helps to improve the controllability of catalysis.
In terms of economic viability, continuous flow methods have lower costs and produce consistently higher-purity products, making them an attractive choice for industrial chemical producers. They are also considered to be “green” methods, which can satisfy the sustainability aims of the fine chemicals industry.
However, companies may be reluctant to utilize this technology despite its benefits due to the need to replace expensive, conventional bulk reactors, requiring significant capital investment in plants. However, the long-term benefits to the chemical industry in terms of cost, product quality, sustainability, and green chemistry can far outweigh the initial financial requirements.
Future Perspectives
The paper has predicted that in the near future, chemical production will shift from utilizing conventional methods to heterogenous catalysis under continuous flow conditions. Benefits for companies include lower costs, high efficiency, high product quality, quicker production times, and increased lot-to-lot product consistency. Novel catalysts can be supported by these techniques.
Single-atom catalysts provide distinct advantages over currently reported catalysts due to their high stability and the requirement for lower amounts of expensive and rare heterogenized catalytic species. Industrial uptake of these catalysts and innovative continuous flow technologies will require a concerted effort by companies and educational establishments to train and employ industrial chemists competent in these methods.
Further Reading
Pagliaro, M, Della Pina, C & Ciriminna, R (2022) Continuous Flow Single-Atom Catalysis: A Viable Organic Process Technology? ChemCatChem e202200768 [online] chemistry-europe.onlinelibrary.wiley.com. Available at: https://chemistry-europe.onlinelibrary.wiley.com/doi/10.1002/cctc.202200768
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.