Jul 1 2016
A team of researchers from the University of Zurich have created a system that would enable swapping of adhesion and stiction (static friction) of a water drop on top of a solid surface. The alteration in voltage is indicated macroscopically in the contact angle between the surface and the drop. This effect can be linked to the alteration in the surface properties on the nanometer scale.
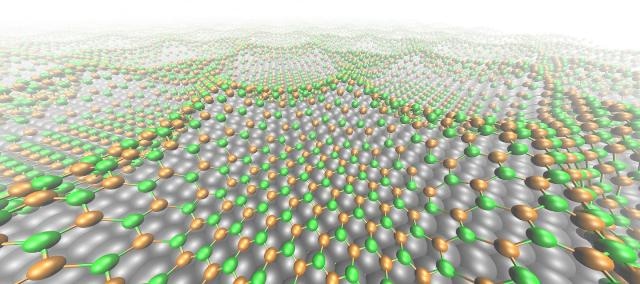 The boron nitride nanomesh superhoneycomb: nitrogen (green), boron (orange), rhodium (grey); distance between honeycombs 3.2 nm. (Photo credit: Marcella Iannuzzi, UZH & Ari Seitsonen, ENS Paris)
The boron nitride nanomesh superhoneycomb: nitrogen (green), boron (orange), rhodium (grey); distance between honeycombs 3.2 nm. (Photo credit: Marcella Iannuzzi, UZH & Ari Seitsonen, ENS Paris)
What enables a gecko to walk across a ceiling upside down? One mechanism which makes this possible is adhesion via the numerous very fine hairs on its feet. This enables the gecko to stick to ceilings and walls. When it moves, it depends on stiction. However, any alteration of stiction and adhesion at macroscopic level is indicated on the nanometer scale via the alteration in the forces applied between molecules and atoms.
How a drop of water touches a honeycomb structure
The team headed by Thomas Greber from the University of Zurich’s Physik-Institut managed to alter the manner in which a drop of liquid sticks to a surface by modifying the electric voltage applied to a water drop. The surface on top of which the drop lies is made up of a material referred to as nanomesh, a single boron nitride layer on metallic rhodium. The structure is in the shape of a honeycomb with the depth of comb at 0.1nm and comb-comb distance at 3.2nm.
Macroscopically, the alteration in electrical voltage is indicated in the alteration of the contact angle between the nanomesh surface and drop. The wetting angle or contact refers to the angle that a drop of liquid takes in relation to the surface of a solid. This angle can be calculated with the help of backlit photographs.
Change in the surface structure alters the contact angle of the drop
On the nanometer scale, the alteration in voltage causes the nitrogen bonds with the rhodium to be substituted by hydrogen-rhodium bonds, which compresses the nanomesh structure. The strength with which the boron nitride’s nitrogen sticks to the rhodium surface is based on its direction to and distance from the next rhodium atom. This establishes the depth of the boron nitride layer and the honeycomb structure.
If the voltage is altered, hydrogen gathers between the rhodium and the boron nitride layers, which makes the honeycomb boron nitride layer to become compressed. Tunneling microscopy can be applied to identify this nanoscopic effect – the alteration in the nanomesh’s surface properties – in the liquid.
To understand and control the interplay between the macro and the nano-world is the real challenge in nanoscience.
Greber
In any case, six orders of magnitude have to be bridged – from millimeters in length (10-3m) to nanometers (10-9m); that is a factor of one million. “Our model system of the electrically switchable nanomesh and a drop’s observable contact angle enables us to access the fundamental phenomenon of the friction of liquids on surfaces more precisely. This should help us solve problems that crop up during lubrication more effectively, for instance.” The study appears on the cover of the latest issue of the Nature journal.
In one aspect of this study, the innovative system is appealing for biology. Using this effect should make it achievable to manipulate the adhesion and movement of cells. Factors such as cell migration or the formation of intricate, multicellular structures with novel scientific methods are likely to be studied as a result.
In the other aspect of this study, technological applications such as capillary pumps, where the height of the capillary can be manipulated via electrical voltage, or micro-capillaries, where the flow resistance can be manipulated, are also feasible.