Jul 21 2016
Fast rechargeable all-solid-state lithium batteries could become one of the next generation of energy storage devices due to their outstanding cycle stability, safety and high energy density. In addition, having higher lithium-ion conductivity of more than 1mS/cm at room temperature, the oxide-based solid electrolyte materials must also have chemical stability.
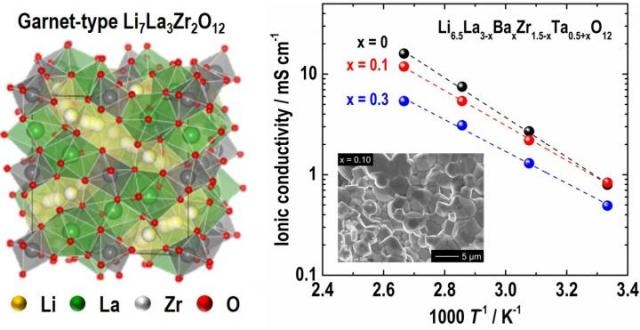 The figure shows crystal structure of cubic garnet-type Li7La3Zr2O12 (LLZO, left) and temperature dependence of ionic conductivity for Ba- and Ta-substituted LLZO with different compositions. Credit: COPYRIGHT (C) TOYOHASHI UNIVERSITY OF TECHNOLOGY. ALL RIGHTS RESERVED.
The figure shows crystal structure of cubic garnet-type Li7La3Zr2O12 (LLZO, left) and temperature dependence of ionic conductivity for Ba- and Ta-substituted LLZO with different compositions. Credit: COPYRIGHT (C) TOYOHASHI UNIVERSITY OF TECHNOLOGY. ALL RIGHTS RESERVED.
These oxide-based solid electrolyte materials have numerous advantages over sulfide-based ones, including ease of handling and chemical stability. However, the creation of a solid-solid interface along with low resistance between the electrode and the solid electrolyte is an additional challenge to obtain enhanced electrochemical performance in solid-state batteries comprising of oxide-based solid electrolyte materials.
Ryoji Inada and his collaborators at the Department of Electrical and Electronic Information Engineering, Toyohashi University of Technology, who participated in this study, created a garnet-type oxide solid electrolyte with high ionic conductivity for an all-solid-state lithium battery. They used this newly developed material to fabricate and test the rechargeable all-solid-state battery.
The researchers systematically examined the impact of alien cation (Ba2+ and Ta5+) substitution in Li7La3Zr2O12 (LLZO, Fig. 1) on the microstructure, crystal phase and the ionic conducting property. The Li concentration was set at 6.5 per chemical formulae in order to stabilize the highly conductive cubic garnet phase, so that the chemical formula of the samples may be stated as Li6.5La3-xBaxZr1.5-xTa0.5+xO12 (LLBZTO).
Consequently, the maximum room temperature conductivity of 0.83 mS/cm was attained in LLBZTO garnet with the Ba content of 0.1 and the Ta content of 1.6. By increasing the Ba substitution level, the Activation energy for LLBZT garnet was monotonically decreased, however, the increased Ba and Ta substitution levels corrupted the conductivity.
The research team also confirmed that the LLBZTO garnet exhibits a broad electrochemical potential window, therefore, a wide range of positive as well as negative electrode materials can possibly be used to develop an all-solid-state battery. They used the aerosol deposition method to fabricate a TiNb2O7 (TNO) film electrode on the LLBZTO garnet and revealed its charge and discharge reaction utilizing a TNO/LLBZTO /Li all-solid-state battery sample.
The study results show that the newly constructed LLBZTO garnet can be utilized as a solid electrolyte material in an all-solid-state battery and help make a very safe rechargeable battery possible for large-scale power resources, although further research is required to improve the performance of solid-state lithium batteries. The research team performed additional investigation to make solid-state lithium batteries real with high energy density.