May 17 2018
Used for thousands of years as a dependable substance, glass is a well-known concept for most. Nevertheless, there is more to glass than what is obvious. Glass is, in fact, an amorphous material with debatably as much in common with a liquid as with the solid most would think it to be.
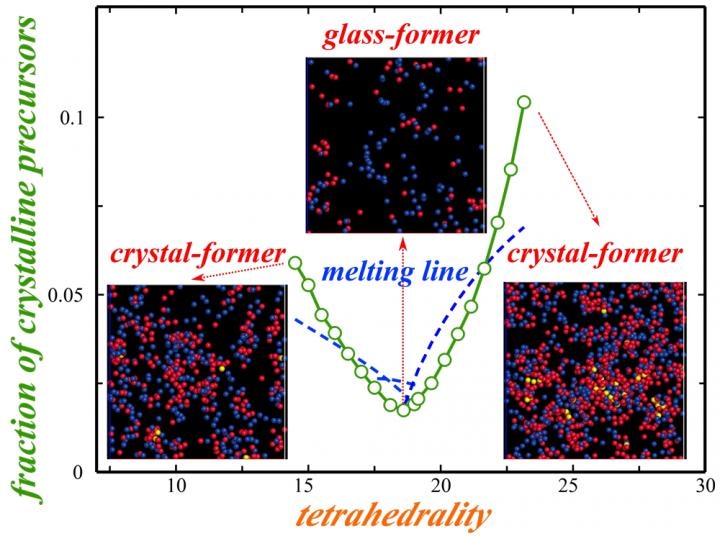 An increase in the glass-forming ability is signaled by a depression of the melting temperature towards its minimum at triple points. (Image credit: 2018 Hajime Tanaka, Institute of Industrial Science, The University of Tokyo)
An increase in the glass-forming ability is signaled by a depression of the melting temperature towards its minimum at triple points. (Image credit: 2018 Hajime Tanaka, Institute of Industrial Science, The University of Tokyo)
Glass comprises atoms fixed into place in a random setup and to those more acquainted with considering the atomic level, "a glass" is the term for any substance in a state that matches this description, leading to a remarkably wide scope.
The propensity for materials to display glassy behavior is referred to as "glass-forming ability" and given the irrefutable success of the well-known silicon-based material, it is easy to comprehend why certain scientists take obtaining a greater understanding of such behavior seriously.
Three researchers based at the Institute of Industrial Science at The University of Tokyo recently examined glass-forming behavior by simulating two model systems whose glass-forming ability could be altered by just one external parameter. Their wide-reaching research findings have been published in Physical Review X.
Glass-forming ability is often influenced by competing effects that suppress the local order that would lead to crystal formation. Our findings show that this behavior is governed by a single parameter that we called the 'thermodynamic interface penalty'.
Hajime Tanaka, Study Corresponding Author
When a material is a mixture of various components, the competition between the different systems attempting to act in their natural way during cooling can result in the creation of a glass. By examining two common systems, the team was able to decouple some of the contributing influences in this process to gain a fundamental know-how of what is occurring.
Our work may provide a general physical principle for controlling glass-forming behavior. The findings could extend to understanding glassy behavior in a variety of systems with competing ordering. This could include structural, magnetic, electronic, charge, or dipolar ordering, which would clearly translate to a very broad range of potential applications down the line.
John Russo, Lead Author
The possibility of using the important findings to manipulate the fabrication and processing of materials such as phase-change materials and metallic alloys opens the door for physics-driven design in various avenues of materials science.