Oct 30 2018
Silicones have a wide array of applications ranging from cosmetics and medical implants to sealants and hydraulic oils to corrosion protection—a critical topic considering the global damages caused by corrosion to the tune of nearly US$3.3 trillion per year.
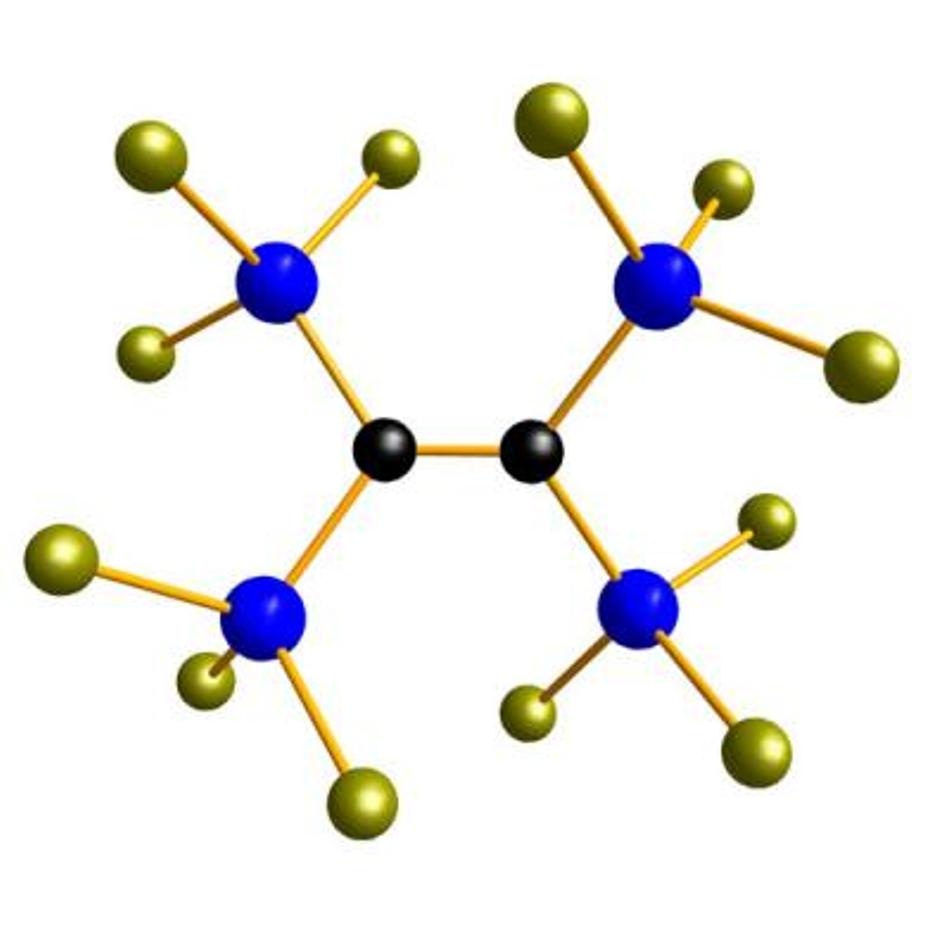 One of the silicon building blocks which permit the design of silicons for various applications. (Image credit: Wagner-group, Goethe-University)
One of the silicon building blocks which permit the design of silicons for various applications. (Image credit: Wagner-group, Goethe-University)
Optimizing silicon-based synthetic materials for particular applications requires made-to-measure chlorosilane building blocks to synthesize and crosslink the long-chain polymers. This has an impact on, for instance, the flow properties and viscosity of the material. Entirely new difficulties are arising in the field of 3D printing, with the help of which products such as individualized running shoes can be produced.
From 1940, the Müller-Rochow Direct Process has been the mainstay of the silicone industry. This process involves using methyl chloride to convert the elementary silicon into methylchlorosilanes at high pressures and temperatures in the presence of a copper catalyst. At present, a working team headed by Professor Matthias Wagner at the Institute of Inorganic and Analytical Chemistry of Goethe University Frankfurt has devised a complementary process with a number of benefits over the Direct Process: It involves using chlorinated hydrocarbons and hexachlorodisilane as starting materials.
“Hexachlorodisilane is already mass-produced for the semiconductor industry and the perchlorethylene (PER) we use particularly frequently is a non-flammable liquid which is so inexpensive that it’s used worldwide as a solvent for dry cleaning,” stated Matthias Wagner. Moreover, the process is performed runs under normal pressure and at room temperature. Instead of a catalyst, only a small concentration of chloride ions is required to activate the process.
“Our process produces highly functionalized organochlorosilanes that are ideal crosslinkers. In addition, their special structure offers excellent possibilities to adjust the mechanical flexibility of the silicon chains as desired,” explained co-inventor Isabelle Georg, whose doctoral dissertation has been sponsored by the Evonik Foundation. Julian Teichmann was also part of the project.
He confirmed that apart from everything, it was the close collaboration between Goethe University and Evonik which had a huge impact on his training: “Regular discussion of our results with Evonik’s industrial chemists opened my eyes from the beginning to economic constraints and ecological requirements. It was fascinating to follow the path from our discoveries in the lab via the patenting procedures to realization on a technical scale in practice.”
The Frankfurt chemists consider that the unique potential of their monomers is due to the fact that they not only comprise of silicon-chlorine bonds but also carbon-carbon multiple bonds. The former plays the role of constructing the inorganic silicon-oxygen chains, and the latter can be linked to formulate organic polymers. This special combination opens innovative routes to inorganic-organic hybrid materials.