Nov 1 2018
Metal-organic frameworks (MOFs)—hybrid materials that stitch metal bits and organic linkers into a mesh-like structure—are widely used for chemical sensing and catalysis, and even as carriers for drug delivery. Those applications could function better if chemists could expand the pores a little more. Now, researchers have demonstrated a method to clip off organic ligands in MOFs to convert micropores into larger mesopores (J. Am. Chem. Soc. 2018, DOI: 10.1021/jacs.8b09682).
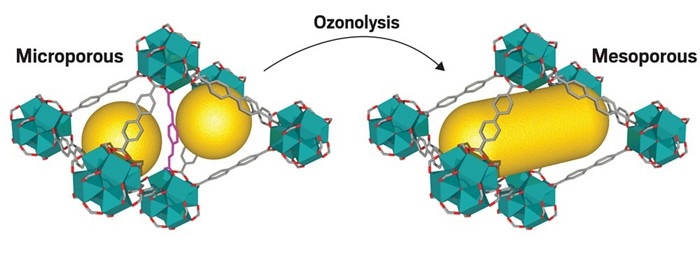 Ozone cuts away certain organic linkers (purple) in a metal-organic framework (left) to make two small pores (yellow) into one larger pore (right). (Image credit: J. Am. Chem. Soc.)
Ozone cuts away certain organic linkers (purple) in a metal-organic framework (left) to make two small pores (yellow) into one larger pore (right). (Image credit: J. Am. Chem. Soc.)
MOFs having mesopores—pores wider than 2 nm—could house larger drug molecules and prevent the gas diffusion that takes place in catalytic applications. Current approaches to increase pore size bank on complex, customized ligands, which are not economical. Just increasing the length of the ligand results in networks of interpenetrating pores that do not expand the size of the gaps. A few approaches exist to customize pore size using various ligands as modulators, but it is difficult to both form a hierarchy of pore sizes and regulate the generation of defects needed for some reactions. Furthermore, some of these approaches use thermal treatments or harsh chemical.
To sidestep these hitches, the trick is to pick ligands that can be selectively cut by reacting with ozone, expanding smaller pores into larger ones, says Vincent Guillerm, a postdoctoral researcher at the Catalan Institute of Nanoscience and Nanotechnology. He and his colleagues designed MOFs from zirconium clusters and two ligands, azobenzene-4,4’-dicarboxylic acid (H2azo) and 4,4’-stilbenedicarboxylic acid (H2sti), both of which are approximately 1.33 nm long. Then, they added ozone into the system, which reacted with the H2sti. Those ligands changed into terephthalic acid and formylbenzoic acid, efficiently severing their links with metallic centers. The H2azo ligands, on the other hand, were not impacted because, unlike H2sti, they do not possess carbon-carbon double bonds that are sensitive to ozone, Gullierm says.
This procedure needed a washing step to eliminate the by-products from the ozone clipping step. The scientists also created a second mesoporous MOF using 4,4’-biphenyldicarboxylic acid and 1,4-phenylenediacrylic acid for the ligands. In this MOF, the products from the cleaved ligands sublimated from the material, thus removing the washing step.
Prior to clipping the ligands, all the pores measured approximately 1.5 nm in diameter. Subsequently, they covered a range of diameters between 2 and 5 nm. The difference arises from the random distribution of the two ligands throughout the material, so certain areas had a higher concentration of one than the other. Scientists hope to gain better control of the distribution, which would also help them narrow the range of pore sizes, says research coauthor Daniel Maspoch, who heads the Supramolecular Nanochemistry and Materials group at the institute.
Besides broadening the pores, clipping the ligands also frees up a few bonding sites that could react with other chemicals. That could prove advantageous for engineering materials other than MOFs. “If you are able to selectively break some bonds inside an object, you can generate some new function in this object,” Maspoch says.
Omar M. Yaghi of the University of California, Berkeley, who is an expert in MOFs, says this work adds a new, imaginative twist to the efforts to enhance MOF performance. “It’s elegant, clever, and its precision is a testament to the increasing utility of reticular chemistry”—the process by which MOFs are stitched together—“in controlling matter on the atomic and molecular level,” he says.