Image Credit: RUDN University.
The new substances were found to have the ability to emit light in the green range, which can be beneficial for medical applications. The study findings were reported in the Molecules journal.
Indolizines are organic substances made of one nitrogen atom and two carbon cycles. They are used in the production of solar panels and dyes, as well as anti-diabetes and anti-tumor drugs. Indolizines are not found in nature but are created in laboratories, often using cycloaddition reactions.
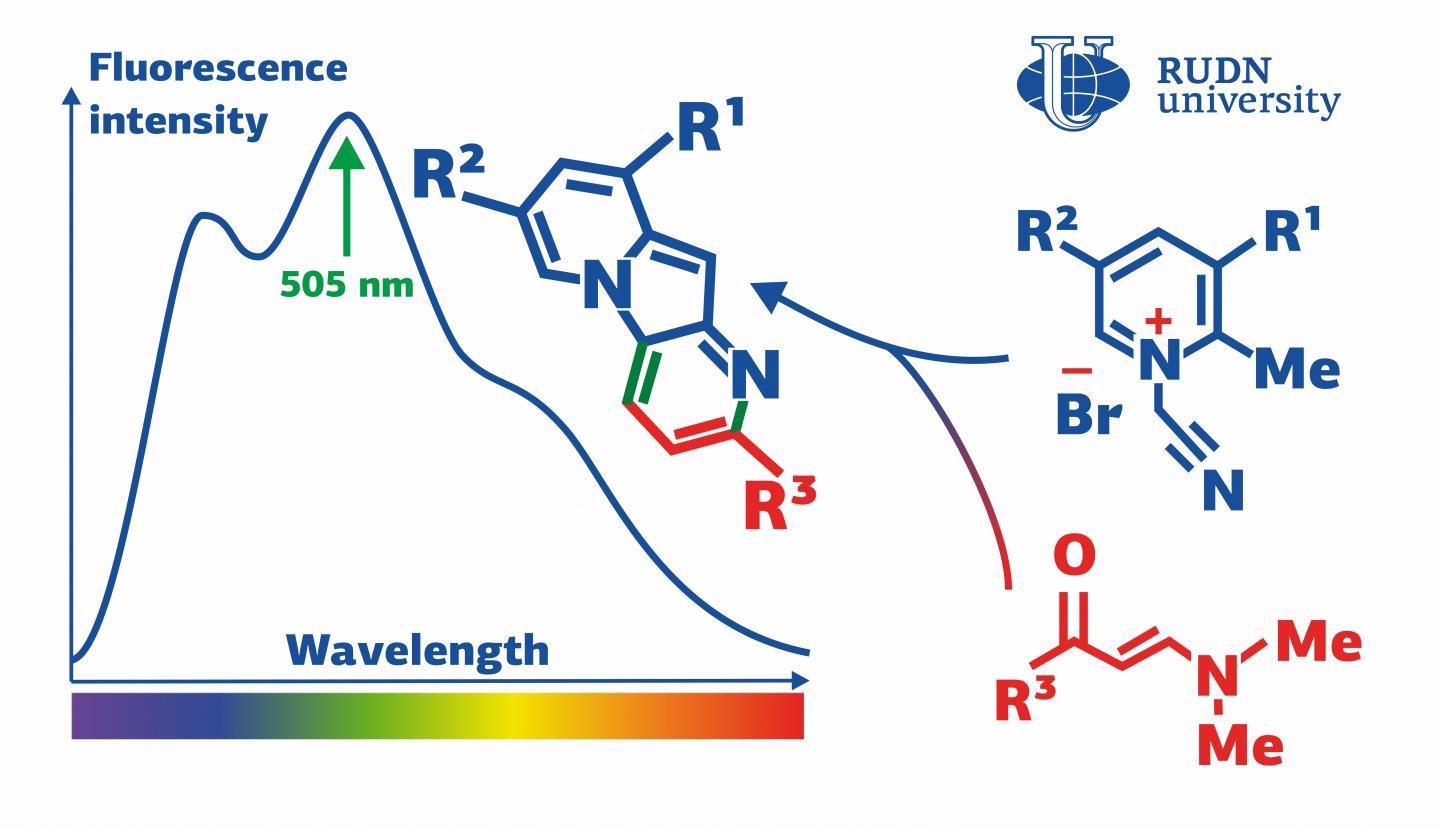
Pyridinium salts—electrically neutral molecules that include positively and negatively charged “poles” balancing out each other—are used in these reactions. The RUDN chemists discovered an unpredicted reaction where, in the place of cycloaddition, a pyridinium salt experiences two other consecutive processes. The final products of this reaction are indolizines that exhibit fluorescent properties.
We found out that pyridinium salts that contain a methyl group bound with ?(2) tend to enter into an unexpected domino reaction with enaminones, and consecutive cycloisomerization and cyclocondensation take place, while the reactions of cycloaddition that are typical for pyridinium salts are not observed.
Alexey Festa, PhD, Senior Lecturer, Department of Organic Chemistry, RUDN University
Chemists from RUDN University, Moscow State University (MSU), and KU Leuven (Belgium) proposed a method based on their previous research works on the reactivity of pyridinium salts. They analyzed the reactions of 1-(cyanomethyl)-2-alkylpyridinium salts with enaminones and selected ideal conditions for the synthesis of indolizines.
The team modified the ratio of the initial components and managed to achieve just a 50% reaction yield. But after using other pyridinium salts, there was an 82% increase in the yield.
The two-stage reaction was found to be of the domino type, where the first stage triggered the second one in the same vessel without the need to add new reagents or any variations in the reaction conditions. The chemists employed X-ray structural analysis to analyze the new indolizines while also focusing on their optical properties.
Of the new indolizines, eight were capable of exhibiting intensive fluorescence (i.e. absorbing light with a specific wavelength and emitting light with a longer wavelength). This mechanism is governed by the excitement of electrons, that is, their movement to a higher energy level under the impact of photons.
This process also brings with it energy release, where a portion of it is emitted as photons with decreased energy and thus increased wavelength. The indolizines produced by the team were specifically effective in absorbing emissions with the wavelength of 403–420 nm, falling in the blue-violet range bordering on the UV light.
The wavelength of the light emitted by the indolizines was found to be 505–528 nm, corresponding to the green range of the spectrum. These properties render indolizines a potential material for making fluorescent tags used to analyze biological objects.
Pyrido[2,3-b]indolizines obtained in the course of our reaction showed certain fluorescent properties. Namely, they emitted green light with a high quantum yield (a parameter that characterizes the efficiency of this process). The lowest yield values amounted to 55-63% and in the case of one new indolizine the yield reached 82%.
Alexey Festa, RUDN University
Journal Reference:
Sokolova, E. A., et al. (2020) Microwave-Assisted Synthesis of Fluorescent Pyrido[2,3-b]indolizines from Alkylpyridinium Salts and Enaminones. Molecules. doi.org/10.3390/molecules25184059.