Image Credit: Wiley-VCH, “Angewandte Chemie.”
The majority of the global energy requirements are still being fulfilled by fossil fuel burning. This adds up to the greenhouse effect in the form of CO2 discharge. To decrease global warming, people should look for possibilities to utilize CO2 as a raw material for basic chemicals.
Furthermore, these could be burned as required. Transformation into liquid fuels would be beneficial since they possess high energy density and are safe to store and transport. But the electrocatalytic formation of products that have two or more carbon atoms (C2+) is very hard.
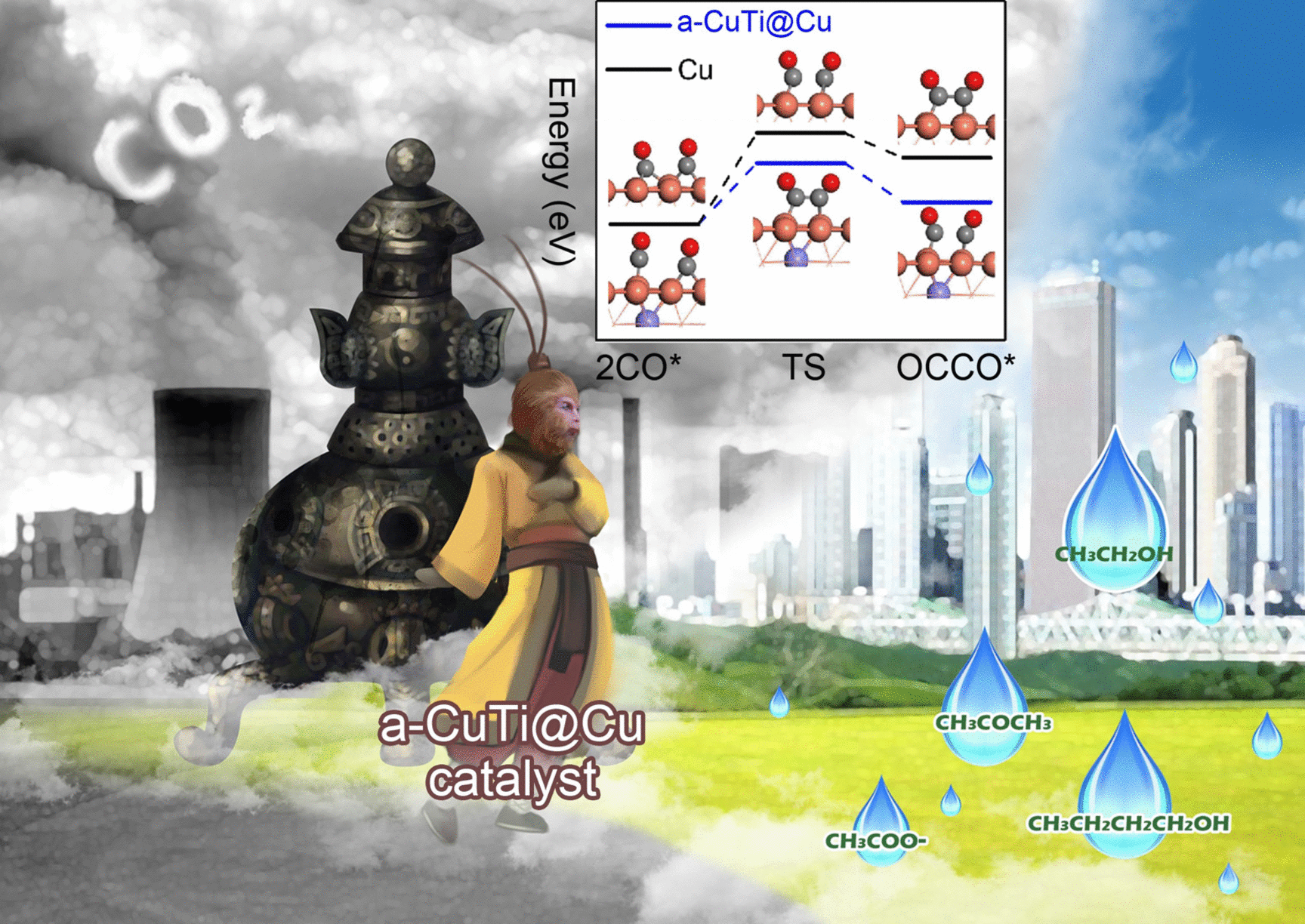
A research group from Foshan University (Foshan, Guangdong), the University of Science and Technology of China (Hefei, Anhui) and Xi’an Shiyou University (Xi’an, Shaanxi), headed by Fei Hu, Tingting Kong, Jun Jiang, and Yujie Xiong, has currently come up with a new electrocatalyst that efficiently transforms CO2 into liquid fuels with several carbon atoms (C2–4). The main products obtained are acetone, ethanol and n-butanol.
The electrocatalyst is made by etching thin ribbons of a copper or titanium alloy with hydrofluoric acid to eliminate the titanium from the surface. This leads to a material called a-CuTi@Cu, which has a porous copper surface on an amorphous CuTi alloy.
It features catalytically active copper centers with exceptionally high selectivity, activity and stability for the decrease of CO2 to C2+ products (complete Faradaic efficiency of around 49% at 0.8 V vs reversible hydrogen electrode for C2–4, and it is stable for a minimum of three months). On the other hand, pure copper foil generates C1 products but barely any C2+ products.
The reaction includes a multistep electron-transfer process through several intermediates. In the latest electrocatalyst, the inactive titanium atoms present beneath the surface play an essential role; they raise the electron density of the Cu atoms on the surface.
This is responsible for stabilizing the adsorption of CO, the main intermediate involved in the formation of multicarbon products. This enables high coverage of the surface with CO and decreases the energy barrier for di- and trimerization of the CO as new carbon–carbon bonds develop.
Journal Reference:
Hu, F., et al. (2021) Ultrastable Cu Catalyst for CO2 Electroreduction to Multicarbon Liquid Fuels by Tuning C–C Coupling with CuTi Subsurface. Angewandte Chemie International Edition. doi.org/10.1002/anie.202110303.