Integrins are one of the major cell receptors present across the cell membrane, responsible for cell adhesion, whether it is cell to cell adhesion or by coupling the extracellular matrix (ECM) outside a cell to the actin of cytoskeleton inside the cell.
These mechanical links are driven by intracellular or extracellular stimuli initiated mechano-transductions. Integrins combine with multi-protein adhesion complexes and promote actin polymerization to form the cellular mechanism that responds to not only the biochemical stimuli but also the physical and topographical properties of ECM. These multi-protein complexes include nascent adhesions (NAs), focal adhesions (FA), fibrillar adhesions, and sliding adhesions.
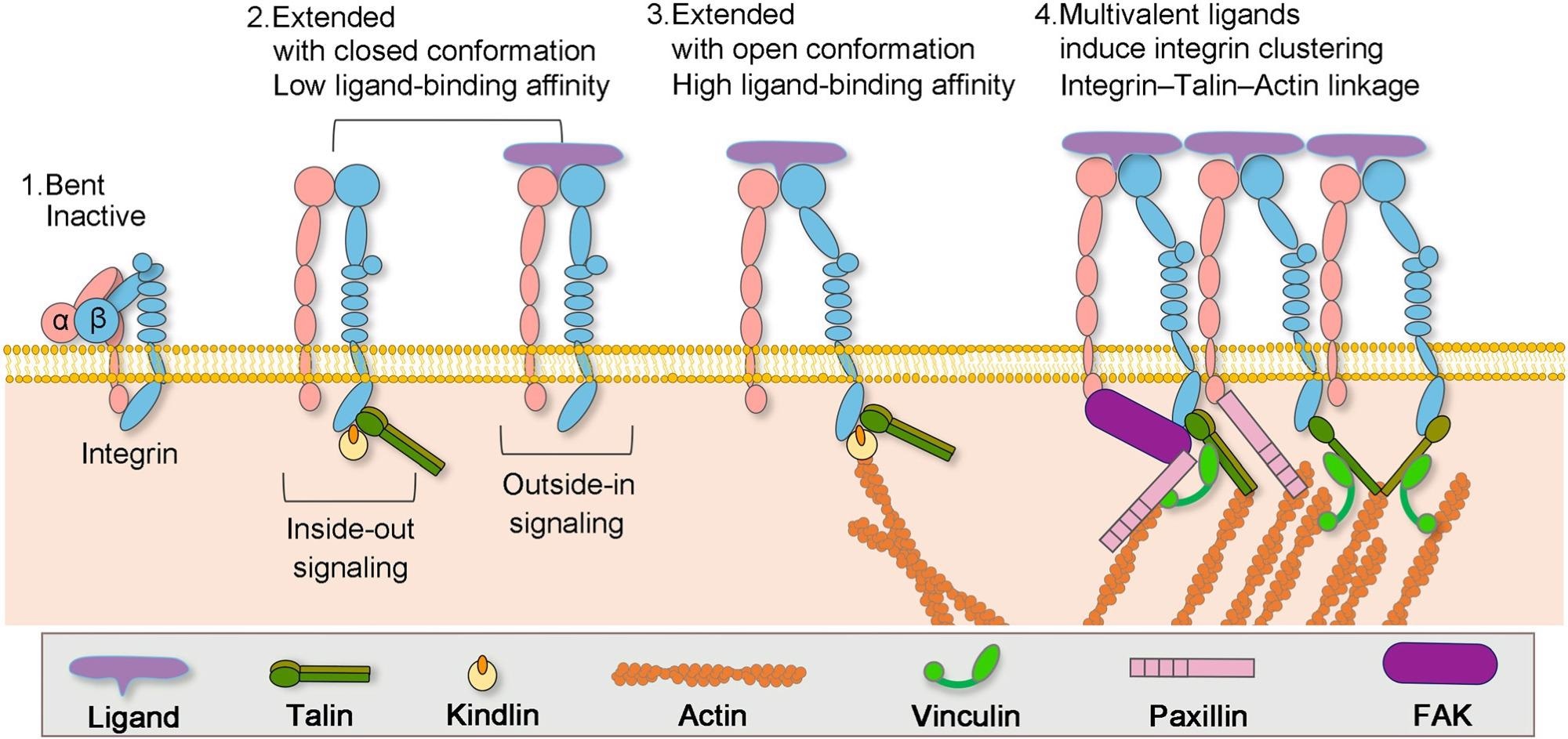
Formation of actin–integrin linkage and integrin activation. Step 1: low-affinity integrin has an inactive, bent conformation. Step 2: inside-out integrin activation by cytoplasmic proteins or outside-in integrin activation via ECM ligands leads to complete extension of the extracellular domains. Step 3: the open, high-affinity activated integrin has the cytoplasmic leg domains separated and links to the actin cytoskeleton via talin. Step 4: multivalent ligand binding induces clustering of integrins. Image Credit: X. Hu, Y. Zhang, Biophysics Review
Other than providing physical coupling, they also facilitate signal transduction between ECM and cell by modulating the signaling pathways of transmembrane protein kinases such as receptor tyrosine kinases (RTK).
Based on the instruction provided by these signals, a cell can execute various cell functions such as cell growth, cell division, cell survival, apoptosis (programmed cell death), cellular differentiation, and blood clotting. The other functions of integrins include acting as receptors for certain viruses such as adenovirus, echovirus, poliovirus, hantavirus, and foot-and-mouth disease.
Integrins are classified into four subfamilies such as collagen-binding integrins, laminin-binding integrins, RGD-binding integrins, and leukocyte integrins. Also, they have two subunits such as alpha and beta. The composition of these subunits varies from cell to cell and species to species, and it determines which protein complex will couple with which ECM molecule.
The ECM is a mesh network consisting of cell-adhesive molecules and signaling molecules. The cell-adhesive molecules facilitate cell cohesion whereas signaling molecules direct cell morphology and behavior.
What are the Factors Affecting Spatial Distribution of Integrins?
The spatial distribution of integrins along with the interactions between integrins and actin cytoskeleton, control the formation and dynamics of integrin-based adhesions. The formation and function of these adhesions include activation, clustering, growth, maturation, and disassembly, which are regulated by the ligand affinity to integrins.
The adhesion molecules are adaptor proteins such as talin and vinculin, and signaling molecules such as paxillin and FAK. Moreover, actin polymerization is also necessary for the formation of NAs.
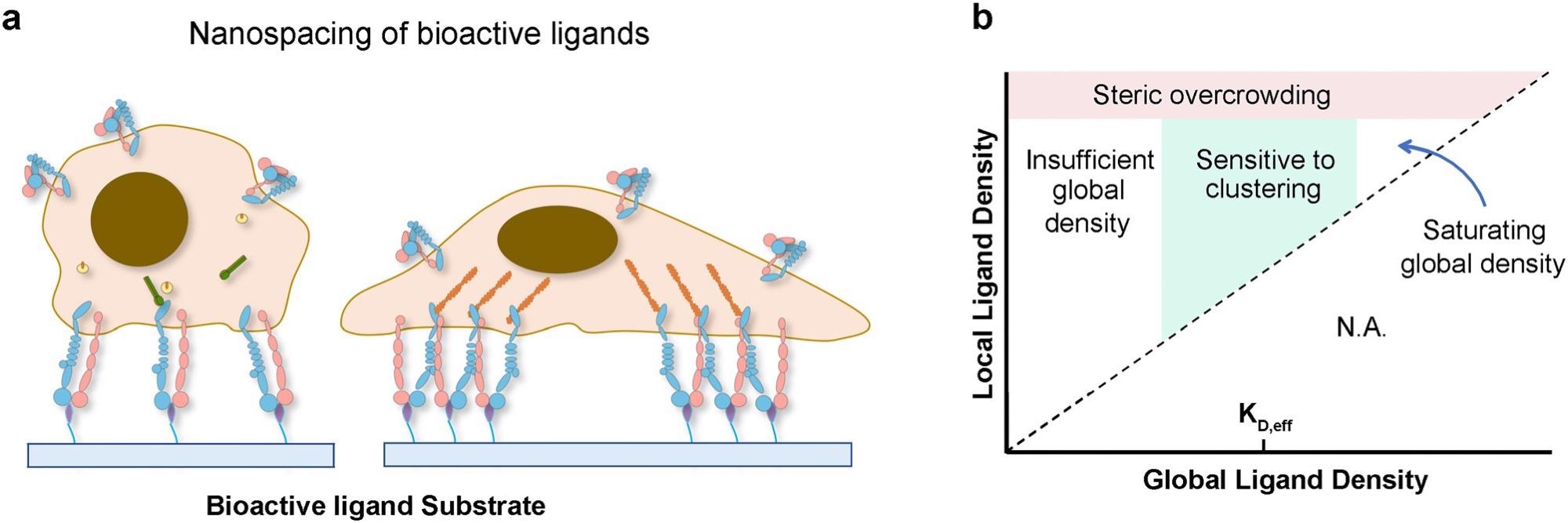
Chemical perturbations in the ECM regulate cell function via integrin: When cells are adherent to biomaterials presenting monovalent ligands, focal adhesion formation and cell spreading are impaired; however, culturing cells on surfaces functionalized with multivalent ligands promotes the formation of adhesion complexes and cell spreading (a). Phase diagram of integrin signaling in various global and local ligand density regimes (b). Image Credit: X. Hu, Y. Zhang, Biophysics Review
Adhesion complexes near the rear of cells and central regions of cells can be disassembled to promote cell migration and spreading. Disassembly of adhesions is primarily facilitated through changes in cytoskeletal tension and tyrosine phosphorylation events. Moreover, several other aspects such as proteolytic activation, microtubule targeting, and actomyosin-based contraction force also contribute to this process.
The adhesion dynamics include the transitions between adhesions such as a diverse range of changes in the morphology, changes in molecular constituents and their locations, correlated cellular functions, and regulated cell-matrix interactions.
Hence, control over a specific integrin isoform distribution leads to the control of adhesion dynamics, whereas the control of adhesion formation leads to control of integrin distribution. Factors that affect the spatial distribution of integrin distribution are non-spacing of bioactive ligands, chemical and mechanical cues, and nano/micrometric topographical cues.
Which Biomaterials can Mediate the Spatial Distribution of Integrins?
Biomaterials such as polymers that can influence the mechanical properties of ECMs, such as elasticity and viscoelasticity on integrin-mediated cell adhesion can effectively regulate the spatial distribution of integrins.
The elastic fibers of ECM mostly consist of elastin and collagen. Elastin fibers provide elasticity, while collagen fibers provide strength and structural support. The team adopted both synthetic elastomers and native elastin derivatives to impart the elasticity of biomaterials.
They investigated the synthetic elastomers such as poly(glycerol-co-sebacate) (PGS), poly(1,8-octane diol-co-citrate) (POC), polyurethanes, poly(e-caprolactone) (PCL), poly(4-hydroxybutyrate) (P4HB), poly(3-hydroxybutyrate) (PHB), and poly(3- hydroxybutyrate-co-3-hydroxyhexanoate) (PHBHHx) for tissue engineering purposes and found them exhibiting promising potentials.
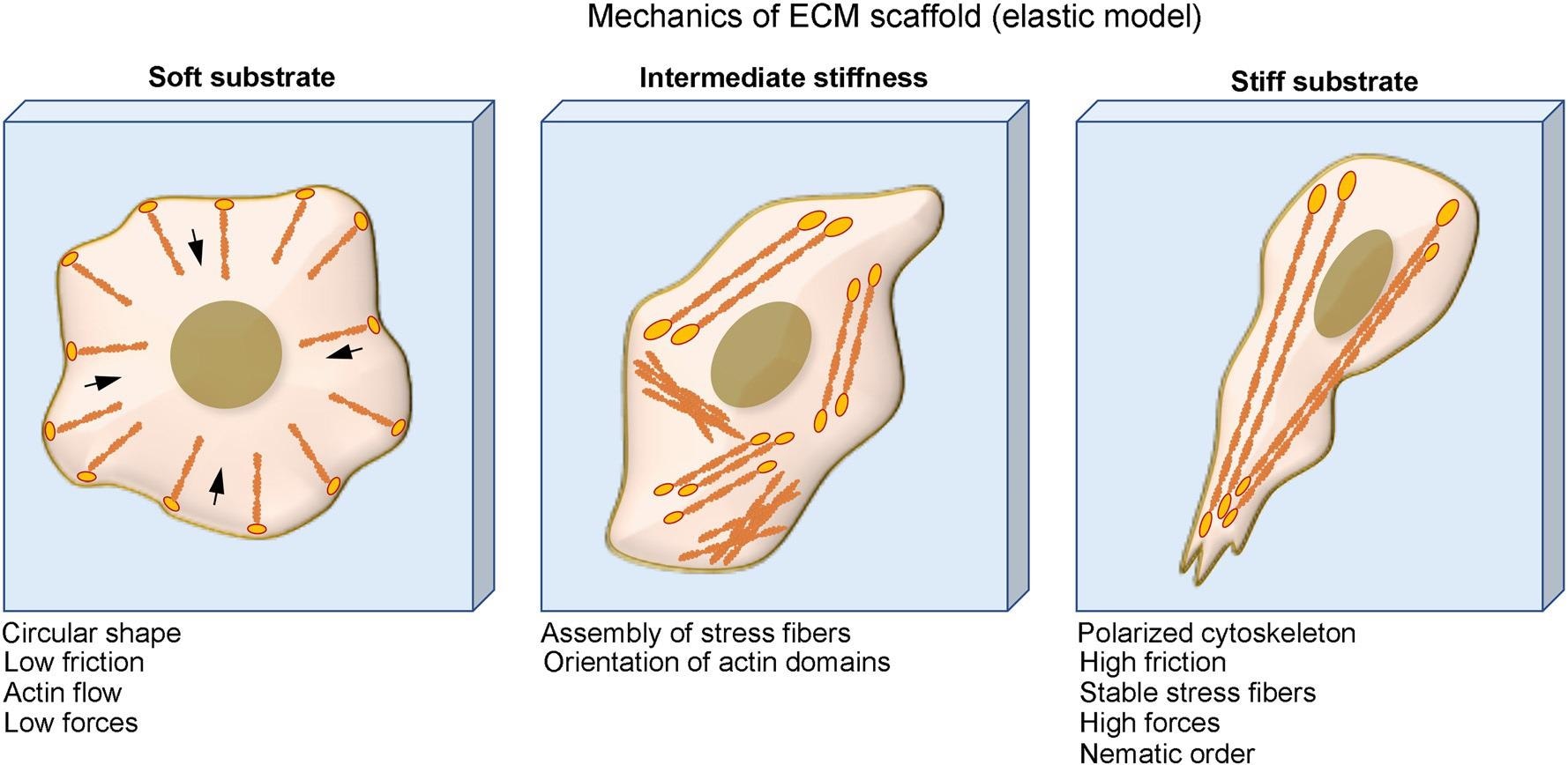
Physical perturbations in the ECM regulate cell function via integrin: Cells on soft substrates have circular shape, low friction, and actin flow. Cells form stress fiber and more organized cytoskeleton when culturing on substrate with intermediate stiffness and cells on stiff substrate have polarized cytoskeleton, high friction, stable stress fibers, and high traction force. Image Credit: X. Hu, Y. Zhang, Biophysics Review
Viscoelastic materials possess a combination of storage of elastic energy in the solid-state and lose mechanical energy in the liquid state. They exhibit stress relaxation in response to a constant deformation and increased creep or strain in response to constant stress.
Polymers that are inert to cell binding and not susceptible to biodegradation are applied as the scaffold of viscoelastic material. The researchers found alginate hydrogels crosslinked by ionic and covalent bonds, alginate hydrogels covalently grafted by polyethylene glycol (PEG) crosslinked by ionic bonds, hyaluronic acid (HA) crosslinked with dynamic covalent hydrazone and collagen I, and artificial protein EPE covalently crosslinked via terminal cysteines and physically crosslinked via midblock domain, P derived from the N-terminal fragment of the rat cartilage oligomeric matrix protein (COMP) are promising materials.
Reference
X. Hu, Y. Zhang, Developing biomaterials to mediate the spatial distribution of integrin, Biophysics Rev. 2, 041302 (2021). https://aip.scitation.org/doi/10.1063/5.0055746
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.