The researchers investigated the superhydrophobicity and the antibacterial activity of the cotton fabric modified with the coating of composite Ag°/rGO against Staphylococcus aureus and Escherichia coli.
Background
Electroconductive fabrics are gaining popularity as a result of their diverse applications. Textiles can be coated with graphene materials (GM) like reduced graphene oxide (rGO) or with carbon nanoparticles such as carbon nanotubes to achieve electroconductivity. Graphene's utility for textile modification is limited due to the difficulty faced in creating stable aqueous dispersions that may be applied to hydrophilic natural fibers.
However, the functional groups in GO make it easier to disperse in water but limit its electrical conductivity, hence GO should be chemically or thermally reduced to rGO. GO chemical reduction frequently requires a high temperature (80–100°C) and a long time, whereas its thermal reduction needs oxygen-free conditions and an even higher temperature.

SEM micrographs of cotton fabric: (a,b) neat, (c) GO-coated, (d) rGO-coated. Image Credit: Makowski, T. et al., Materials
There is currently an increasing demand for smart and intelligent clothes, such as cotton cloth, which are electroconductive and can store or communicate energy. Antimicrobial action can eradicate microbial colonization and lower the risk of infection in such applications. The antibacterial activity of GM particles relies on their adsorption ability, dispersibility, sharp edges, and the number of corners.
Nonetheless, very few studies have documented the cotton fabric modification via chemical reduction of GO and the growth of Ag. Also, the antibacterial characteristics of the fabric treated only with rGO are not yet investigated.
About the Study
In the present study, GO was coated on a cotton fabric based on a padding strategy and subsequently thermally reduced to rGO by using L-ascorbic acid to make the cloth electroconductive. With cyclic voltammetry, Ag° particles were electrochemically placed on the rGO-coated electroconductive cloth, allowing the redox processes to be monitored. The disk diffusion method was used to assess the antibacterial activity of the modified cotton fabric against Gram-negative E. coli bacteria and Gram-positive S. aureus.
The fabric's surface electrical resistivity (R) was tested using the four-wire method, and water contact angles (WCA) were evaluated to test water wettability. The antibacterial characteristics of the fabric samples were examined using the antibacterial activity assessment of textile materials i.e., agar diffusion test against E. coli ATCC 8739 and S. aureus ATCC 6538. The modified cloth fibers were also examined under a scanning electron microscope (SEM) to assess bacterial colonization.
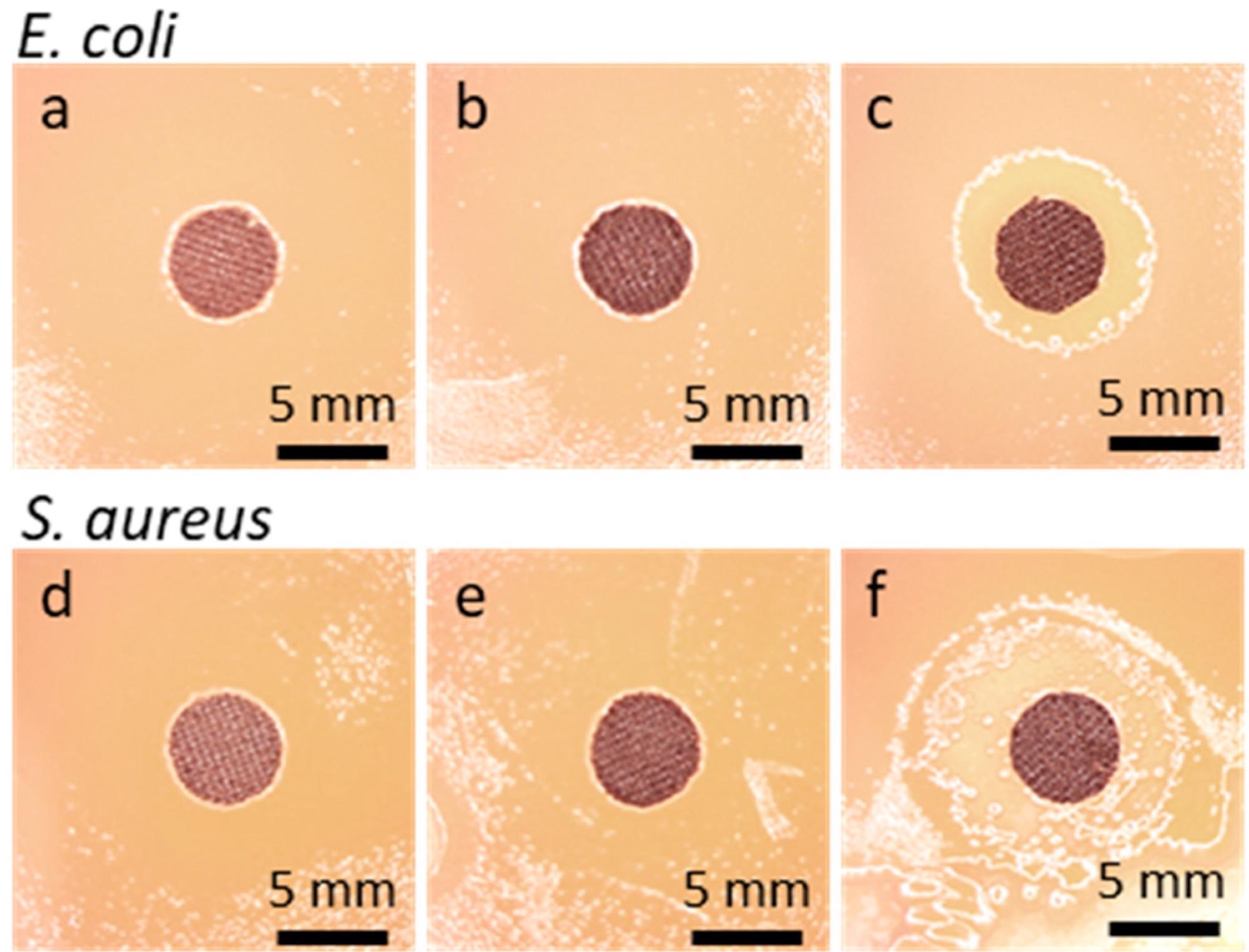
Disk specimens of cotton fabric on bacteria inoculated agar after 48 h incubation: E. coli (a) GO-coated, (b) rGO-coated, (c) Ag°/rGO-coated; S. aureus, (d) GO-coated, (e) rGO-coated, (f) Ag°/rGO-coated. Image Credit: Makowski, T. et al., Materials
Observations
The results showed that the fabric became electrically conductive when the amount of GO placed on it was reduced. R decreased dramatically when the hot stage temperature reached 220°C, but it leveled off within 1 minute at 0.1 MΩ/sq at 220°C. The presence of a conducting 3D network of rGO over the surfaces of fiber was demonstrated by an R of 5.4 MΩ/sq.
On the rGO-coated surfaces, Ag+ ions were reduced, along with the nucleation and development of Ag particles, as inferred from SEM micrographs. The Ag content was determined to be 0.4 wt.% based on TGA analysis. After the electrochemical alteration, the fabric's R remained constant at 5.35 MΩ/sq. With a WCA of 161°, the Ag/rGO-modified cloth was found to be superhydrophobic.
It was observed that the fabric gained electrical conductivity as a result of the reduction of GO, allowing for cyclic voltammetry-based electrochemical deposition of Ag° particles. The coating of the combination of Ag°/rGO composite imparted the fabric antibacterial activity and superhydrophobicity against the studied bacterial strains, S aureus and E coli. It was seen that the coated fabric did not possess antibacterial activity before or after the GO reduction.
Only the Ag°/rGO composite coating gave the cloth antibacterial characteristics. Bacterial film was not identified on Ag°/rGO-modified fibers, and bacterial growth inhibition zones were discovered surrounding the material specimens.

SEM micrographs of bottom surfaces of cotton fabric specimens removed from agar after 48 h incubation: E. coli (a) neat fabric, (b) GO-coated, (c) rGO-coated, (d) Ag°/rGO-coated, S.aureus (e) neat fabric, (f) GO-coated, (g) rGO-coated, (h) Ag°/rGO-coated. Image Credit: Makowski, T. et al., Materials
Conclusions
In conclusion, this study demonstrated the antibacterial activity of a cotton fabric modified with Ag°/rGO composite coating. The coated fabric did not have antibacterial action before or after the reduction of GO and the antibacterial capabilities were only imparted to the fabric by the Ag°/rGO coating, as evidenced by bacterial inhibitory growth zones around the investigated specimens as well as the non-existence of bacterial films on the fibers. In addition, the Ag°/rGO-modified fabric became superhydrophobic, with a WCA of 161°.
Overall, the authors demonstrated that the acquired results on the composite Ag°/rGO fabric coating are promising for the development of novel antibacterial materials.
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.