Gelatin is a protein compound that is produced by native collagen digestion. Collagen is found in the skin, cartilage, and bones of animals and fish, with the most common industrial sources of gelatin pork and the bones and skin of bovines and pigs. Factors that affect the properties of gelatin are its chemical composition and the raw material from which it is obtained.
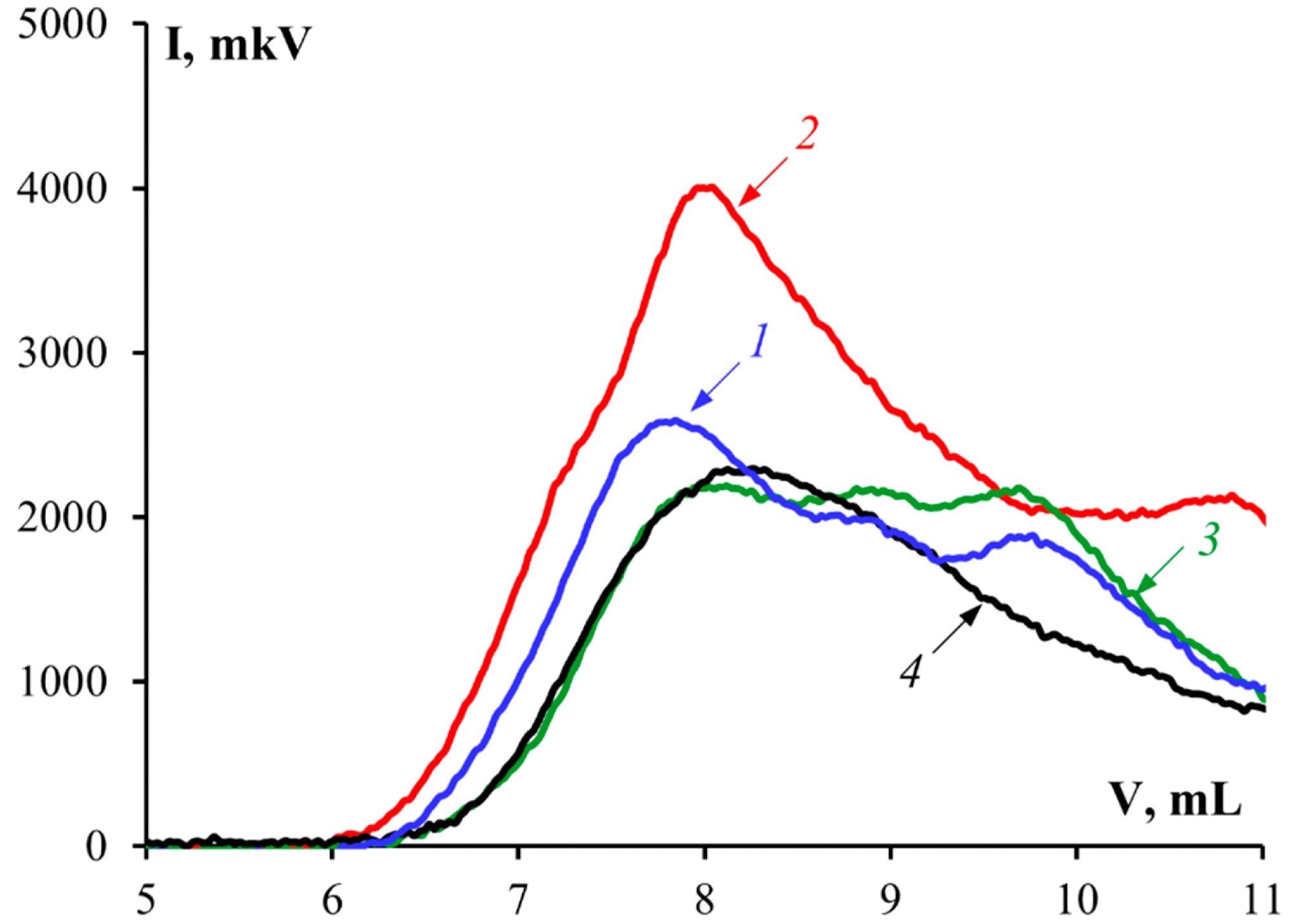
Chromatograms of gelatin samples: 1—G1, 2—G2, 3—G3, 4—G 7041. Image Credit: Derkach, S.R. et al., Polymers
The highly organized, interconnected collagen fiber, which is an endless, asymmetric network of tropocollagen units, transforms into simpler structural units. These simple units possess a lower degree of internal order. The degree of collagen conversion depends on pretreatment and the extraction process conditions (time, PH, and temperature.)
Conventional production of gelatin from collagen involves acid or alkali reactions. Two types of gelatin are produced depending on the type of pretreatment: Type A gelatin (acidic pretreatment) and Type B gelatin (alkaline pretreatment.)
Conventional production methods using acidic or alkaline pretreatments possess significant drawbacks. Methods have long processing times, use substantial amounts of water, and require harsh chemical reagents. Moreover, processes lead to copious amounts of waste, which causes environmental problems.
Producing Gelatin from Raw Fish Materials
Producing gelatin from fish materials (for instance, bones, scales, and skin) requires a different process than conventional acid and alkaline pretreatment and processing. Mild and sparing processes are needed which have a short extraction time and weakly acidic environment. Moreover, a harsh temperature regime that causes the cleavage of gelatin α-chains is not used.
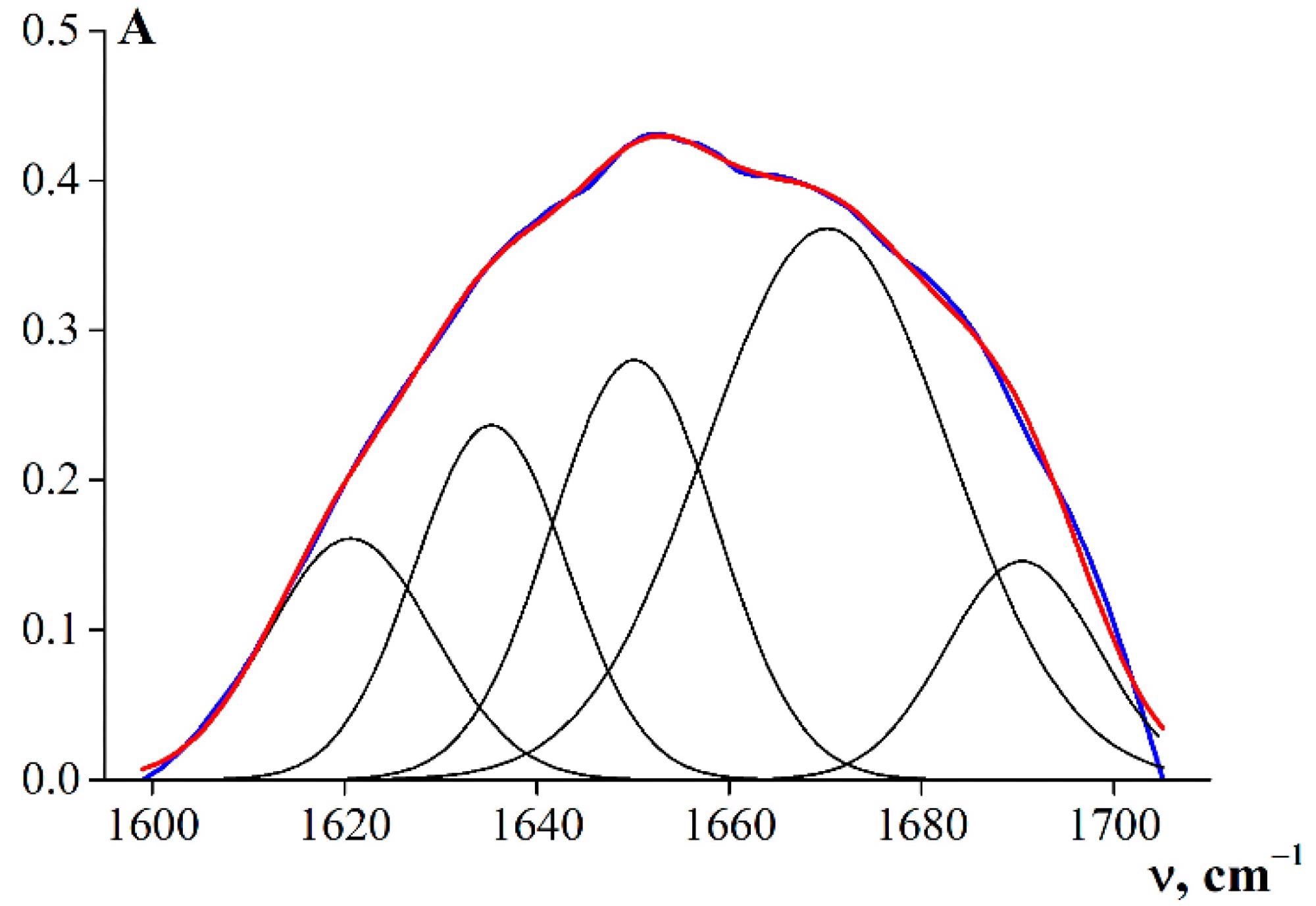
Decomposition of the band components of the amide I band using a Gaussian distribution (R2 = 0.99). Image Credit: Derkach, S.R. et al., Polymers
One method for improving the process of producing gelatin from raw fish materials is the use of enzymes. Enzymes possess high specificity, leading to targeted effects on specific bonds in the collagen matrix. Additionally, they improve digestion rates. Natural enzymes are typically used for this process, including plant proteases such as papatin, and animal proteases including trypsin and pepsin. Another enzyme typically used is Pancreatin, which is a commercial food-grade enzyme.
As well as animal and plant enzymes, microbial enzymes have been utilized in the production of gelatin from fish products. An advantage of using microbial, rather than plant or animal enzymes, is their enhanced catalytic activity and their stability and action across a wider range of pH and temperature.
For instance, studies have demonstrated that Alcalase 2, 4 L significantly accelerates the gelatin extraction process. Additionally, compared to papain, protosubtilin possesses enhanced collagenase activity at a temperature range close to room temperature.
Using Waste Materials for Gelatin Production
In the Northern Basin, commercial fishing activities have increased in recent years due to the demands of a growing global population. This increase in fishing has led to significant interest in the production of gelatin from these stocks, in particular Atlantic cod. Catches have increased by 4% in recent years, to an annual yield of approximately four hundred thousand tons. The production of skinned fillets for the food industry produces copious amounts of collagen-containing materials that can be utilized for producing gelatin.
The Study
The study in Polymers has categorized the production of gelatin produced from the skin of Atlantic cod using enzymatic treatments. The authors have stated that issues related to increasing product yield and decreasing the time needed for commercial production of gelatin from raw fish materials are largely unresolved in current literature. Preserving the functional and physicochemical properties of fish gelatin whilst improving the technology is a key factor in research, which the authors have attempted to address.
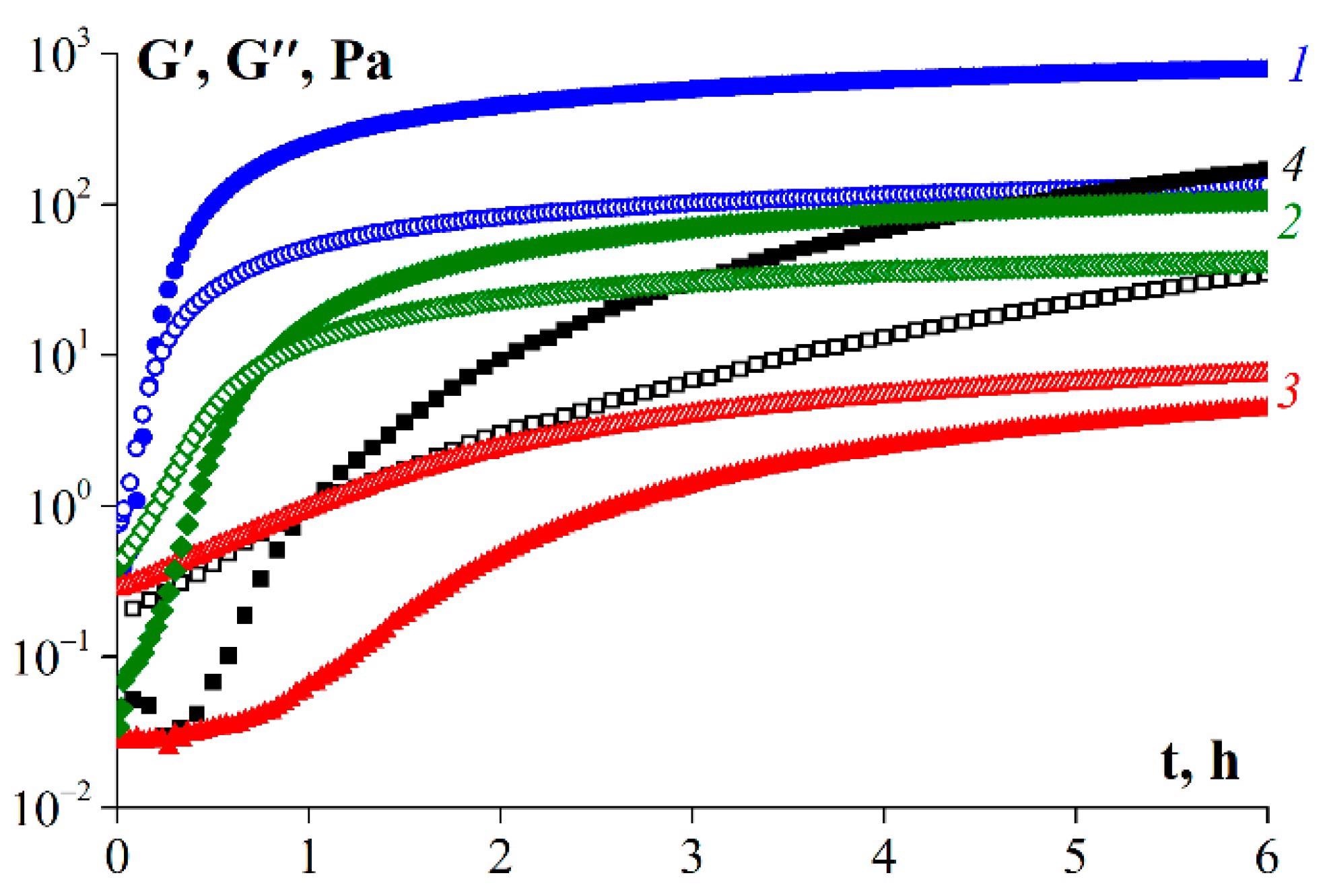
Gelation kinetics of gelatin samples: 1—G1, 2—G2, 3—G3, 4—G 7041. T = 6 °C, ω = 6.28 rad/s, γ = 1%. Gelatin concentration was 10 wt.%. The initial temperature of the samples was 25 °C. Image Credit: Derkach, S.R. et al., Polymers
The authors chose pancreatin and the enzyme preparation protosubtilin, which are both widely available commercially, for the preparation of enzyme-based gelatin. Pancreatin and protosubtilin were chosen because of their optimal activity at close to room temperature (~40oC) and the fact that they do not cause protein denaturation and the loss of functional properties in gelatin. Energy requirements for gelatin production are reduced by their use.
Additionally, these enzymes possess optimal activity at neutral pH, reducing the need for reagents to regulate pH, and consequently, produce less waste. Furthermore, they are safe to use and can produce food-grade gelatin. Thus, the focus of the study was improving the technology for producing gelatin from fish waste, reducing the time needed to extract it, and increasing yields without losing the gelatin’s functional properties.
Results of the study demonstrated a reduction in time duration for extraction by 40%, and an increased yield of 58-60%, compared with 51% without enzymatic treatment. Both pancreatin and protosubtilin produced gelatin with a lower molecular weight. Pancreatin produced gelatin with the poorest rheological properties.
The authors concluded that using protosubtilin is economically beneficial, reducing extraction time, and increasing yield whilst producing gelatin with similar or superior functional properties and qualities as commercially available fish gelatin. Moreover, the number of reagents needed is reduced, which means the proposed method can be used in environmentally friendly technologies.
Further Reading
Derkach, S.R. et al. (2022) Characterization of Fish Gelatin Obtained from Atlantic Cod Skin Using Enzymatic Treatment [online] Polymers 14(4) 751 | mdpi.com. Available at: https://www.mdpi.com/2073-4360/14/4/751
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.