The production of electric vehicles (EVs) is on the rise in an attempt to curb the dependency on fossil fuels that contribute to an increase in carbon emissions and thereby global warming and pollution. Hence, developing batteries, especially LIBs, has become crucial.
Graphite, with its short reduction voltage yielding excellent cyclability, is commonly the anode material used in LIBs. However, the power density for modern batteries is insufficient due to the limited passageway of lithium ions. Methods to improve the graphite anode’s performance rates include surface doping, carbon coating, and graphite spheronization.
Pitch is a residue that is a result of heat treatment and subsequent distillation of coal tar or petroleum fractions. It consists largely of aromatic hydrocarbons and is solid at room temperature but has a wide softening range rather than a definite melting temperature. Additionally, pitch coatings are advantageous in imparting adequate mechanical strength to regulate high conductivity and volumetric expansion, while being cost-effective. Therefore, they have gained attention as a source of carbon coating.
About the Study
In the present study, the team prepared samples of pitch-coated graphite from natural FG and pitch, which were blended in weight ratios of 80:20, 85:15, 90:10, and 95:5 for 30 minutes through centrifugal mixing. Characterization of the samples was carried out for true density, which was computed by a He pycnometer. Surface crystallinity was measured using Raman spectroscopy, and particle morphology and size distribution were determined using scanning electron microscopy (SEM) and laser diffraction, respectively. Other parameters like powder resistivity, packing density, and powder conductivity were also determined.
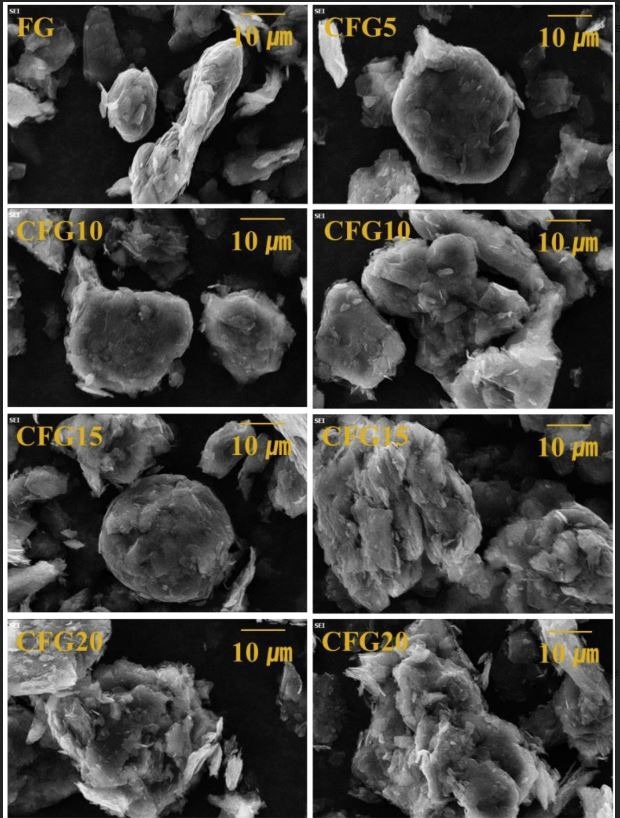
SEM images of FG, CFG5, CFG10, CFG15, and CFG20. Image Credit: Kim, B et al., Materials
To assess the electrochemical attributes, electrodes were made using styrene-butadiene rubber (SBR) and carboxyl methyl cellulose (CMC) as binders. The electrolyte selected was 1 M LiPF6 dispersed in a 1:1 by volume mixture of diethyl carbonate (DEC) and ethylene carbonate (EC), separated by polyethylene (PE). Electrochemical parameters such as the galvanostatic discharge and charge were tested in between a potential of 0.01-1.5V. The performance rate was calculated by lithiating the coin cells at a rate of 0.1C and then delithiating at different rates in the range of 0.1C to 5C. The electrochemical impedance spectroscopy (EIS) was carried out at a 10 mV amplitude with a frequency ranging from 100 kHz-0.01 Hz. The diffusion coefficient was assessed via cyclic voltammetry (CV) ranging from 0.01-1.5V.
Results
The observations showed that 5 wt% pitch coating on FG resulted in a more uniform surface and improved spheroidization as compared to FG without pitch coating, thus enabling a lithium-ion diffusion pathway. Furthermore, pronounced aggregation was observed with rising pitch content, resulting in a rougher particle surface. The particle size distribution exhibited a rapid increase from 15 wt% pitch because of pitch aggregation. However, a substantial reduction in the specific surface area of the pitch-coated samples resulted in a smooth and clean surface.
The first initial coulombic efficiency (ICE) was observed to be higher with 5 wt% pitch coating (84.07%), as compared to that of FG (80.49%). On the other hand, pitch content higher than 5 wt% exhibited lower ICE values than that of FG. In comparison to FG, the discharge capacity retention of FG with 5 wt% pitch coating was 1.9 and 8.8 times higher at 2 C and 5 C whereas higher pitch content exhibited lower capacity retention values. The cycling stability deteriorated with the formation of a graphene layer due to lithium intercalation and deintercalation. The graphene layer formation was inhibited with the addition of pitch.
Conclusions
In summary, the results showed that a uniform pitch coating makes the graphite particles more spherical, improving the ICE. However, at 10 wt% and higher, pitch aggregation occurred resulting in a non-uniform surface. The enhancement in retention capacity could be attributed to the amorphous structure of the carbon pitch providing a lithium-ion pathway thereby improving the rate of performance.
Likewise, the rolling process orients the graphite particles parallel to the copper current collector. The diffusion pathway is extended since the graphite particles are oriented perpendicular to the diffusion direction, thus making the diffusion of lithium ions difficult. A uniformly coated pitch imparts a spherical shape, inhibits particle orientation while rolling, and results in a higher rate of performance.
References and Further Reading
Kim, B.-R.; Kim, J.-H.; Im, J.-S. Effect and Mechanism of Pitch Coating on the Rate Performance Improvement of Lithium-Ion Batteries. Materials 2022, 15, 4713. https://www.mdpi.com/1996-1944/15/13/4713
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.