May 1 2018
Researchers from the Kyoto University have reached a step closer to developing porous materials that can change and maintain their shapes—an attribute called shape-memory effect.
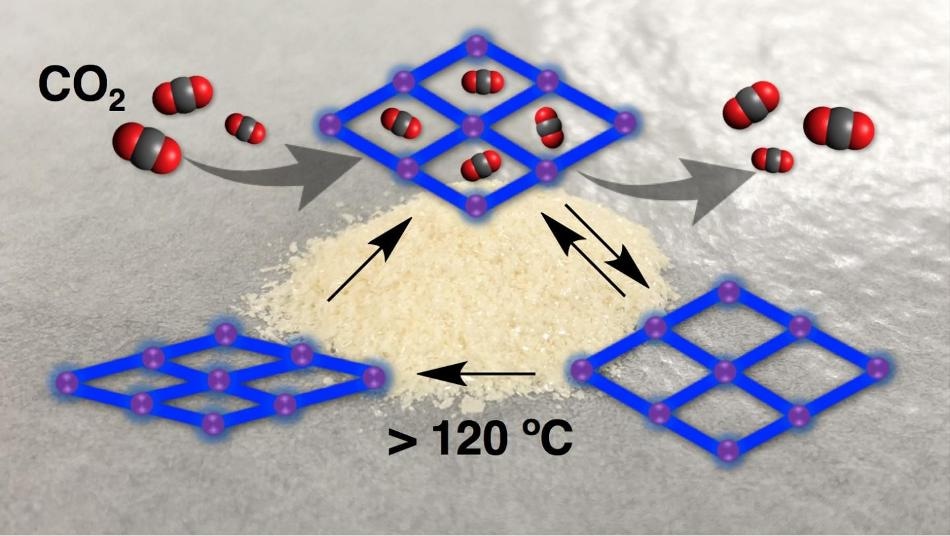 Researchers documented how a porous material can change and retain its shape, even after absorbing and releasing carbon dioxide. Here, the crystal’s pores remain open after releasing carbon dioxide but can be collapsed when heated. (Image credit: Kyoto University iCeMS)
Researchers documented how a porous material can change and retain its shape, even after absorbing and releasing carbon dioxide. Here, the crystal’s pores remain open after releasing carbon dioxide but can be collapsed when heated. (Image credit: Kyoto University iCeMS)
Shape-memory materials have been applied in various fields. For instance, they can be implanted in the human body and then stimulated to change shape for a particular function, for instance, to function as the scaffold for bone tissue regeneration. In certain materials, such as metal alloys and ceramics, the shape-memory effect is well documented. However, in the case of crystalline porous materials, it is uncommon and not so well understood.
At present, Susumu Kitagawa from Kyoto University’s Institute for Integrated Cell-Material Sciences and his collaborators from Japan, Ireland, and the United States have confirmed a shape-memory effect in a flexible metal-organic material—only the second such instance reported ever. Their findings have been reported in the Science Advances journal.
A mixture of zinc nitrate hexahydrate and chemicals was dissolved in a common solvent known as dimethylformamide at 120 °C for 24 hours to develop crystals. The researchers adopted an X-ray method known as single-crystal X-ray diffraction to investigate the structure of the crystals. They discovered that the crystals were made of slightly distorted paddlewheel-shaped lattices, which were composed of central zinc ions connected to surrounding organic molecules. The porosity of this “alpha phase” of the crystal was 46%, indicating that 46% of its volume was free for receiving new molecules—the feature that renders porous materials appropriate for a range of applications.
When the alpha crystal was heated by the researchers for 12 hours at 130 °C in a vacuum, the crystal became highly dense, its lattices turned highly distorted, and its porosity was decreased to just 15%. This phase of the crystal was named by the team as its beta phase.
Then, the team added carbon dioxide to the crystal at a temperature of −78 °C. The pores of the crystal adsorbed carbon dioxide, and the shape of the crystal changed to less-distorted lattices when compared to those in the beta phase. The free volume available for receiving guest molecules increased to 34%. Upon adding and removing carbon dioxide to and from the crystal over 10 consecutive cycles, they discovered that it maintained its shape. The team termed this phase of the crystal its “shape-memory” gamma phase.
The addition of carbon monoxide or nitrogen under different temperatures also stimulated the conversion of the crystal from the beta to the gamma phase.
The researchers were successful in reversing the gamma phase of the crystal back to its beta phase by heating it at a temperature of 130 °C in vacuum for 2 hours. To go back to the alpha phase, the crystal’s gamma phase was soaked in dimethylformamide for five minutes.
The researchers’ investigations of the crystal enabled them to have an in-depth knowledge of the change in its function in tandem with structural changes. The team remarked that their study could offer the basis for the development of more samples of shape-memory porous materials.