Image Credit: Pavel Odinev/Skolkovo Institute of Science and Technology.
Hydrogen hydrates offer a promising solution for the storage and transportation of hydrogen—the most eco-friendly fuel. The study was published in the Physical Review Letters journal. Ice is an extremely complex substance that has numerous polymorphic changes. These changes continue to grow in number as investigators make new discoveries.
The physical characteristics of ice also differ considerably—for example, hydrogen bonds turn out to be symmetric at extreme pressures, rendering it impossible to identify a single water molecule, while low pressures induce proton disorder, putting water molecules in several potential spatial orientations inside the crystal structure.
The ice around the environment, such as snowflakes, invariably has disordered protons. Ice can integrate molecules of chlorine, xenon, methane and carbon dioxide, as well as create gas hydrates. These gas hydrates have a structure that is usually different from pure ice. The large bulk of the Earth’s natural gas occurs in the form of gas hydrates.
Chemists from the United States, China and Russia targeted hydrogen hydrates in the latest study. Gas hydrates attract a great deal of interest for theoretical studies and also for practical applications, like the storage of hydrogen. If hydrogen is stored in its natural state, it poses an explosion risk, while compressed hydrogen has a relatively low density. This is the reason why investigators are searching for low-cost hydrogen storage solutions.
This is not the first time we turn to hydrogen hydrates. In our previous research, we predicted a novel hydrogen hydrate with 2 hydrogen molecules per water molecule. Unfortunately, this exceptional hydrate can only exist at pressures above 380,000 atmospheres, which is easy to achieve in the lab, but is hardly usable in practical applications. Our new paper describes hydrates that contain less hydrogen but can exist at much lower pressures.
Artem R. Oganov, Professor, Skolkovo Institute of Science and Technology
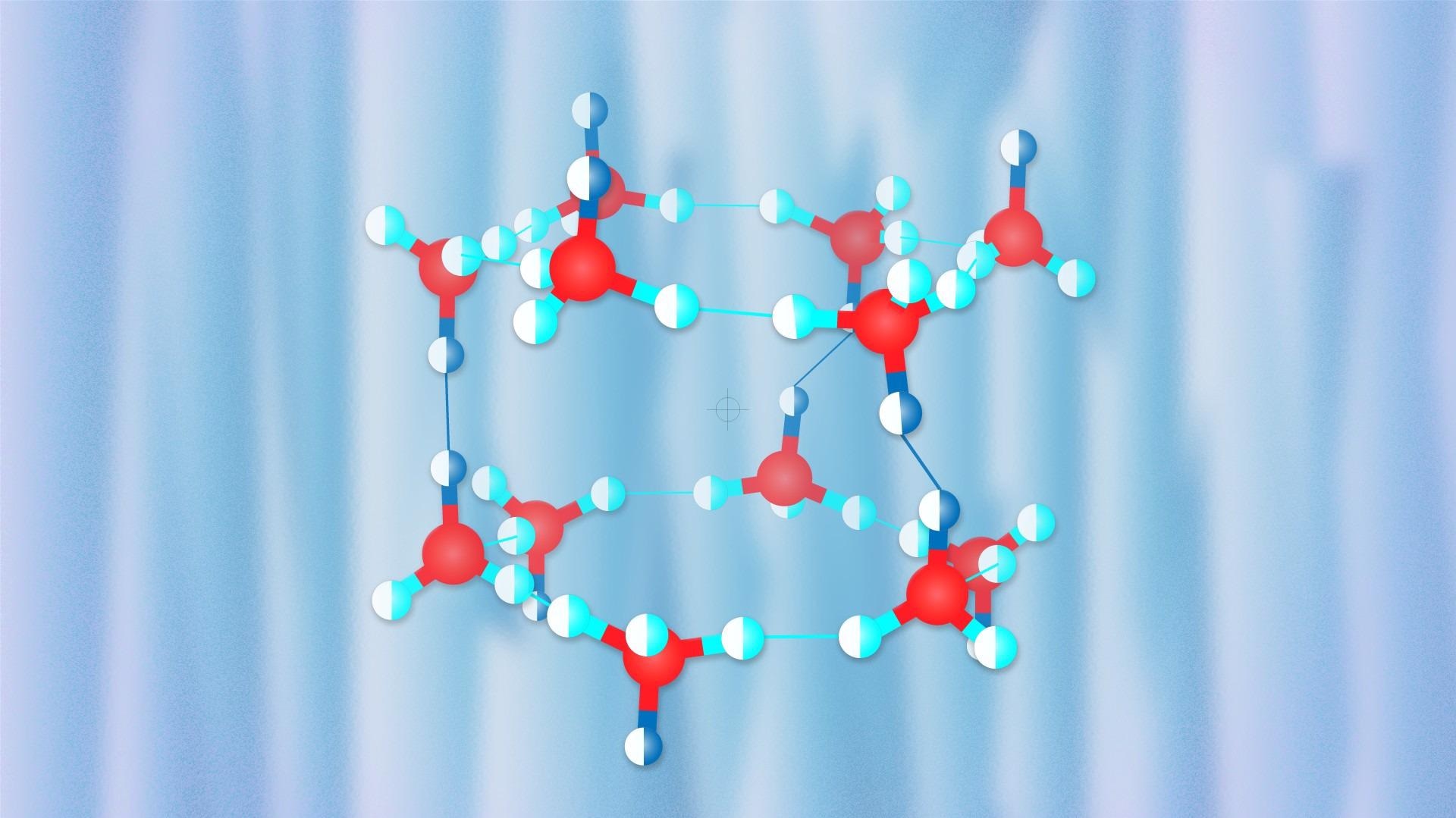
In hydrogen hydrates, the crystal structure strongly relies on pressure. The crystal structure has large cavities at low pressures, and according to Oganov, these cavities look like Chinese lanterns, each accommodating the molecules of hydrogen.
The structure becomes denser as the pressure increases and more hydrogen molecules are closely packed into the crystal structure, albeit their degrees of freedom become considerably fewer.
In their study published in the Physical Review Letters journal, the researchers from the Carnegie Institution of Washington in the United States and the Institute of Solid State Physics in Hefei, China—headed by Alexander F. Goncharov, a Professor from both these institutions—conducted experiments to investigate the properties of numerous hydrogen hydrates and identified a strange hydrate with three water molecules for every hydrogen molecule.
The research group, headed by Professor Oganov, employed the USPEX evolutionary algorithm designed by Oganov and his students to figure out the structure of the compound responsible for its unusual behavior.
The team replicated the conditions of the experiment and discovered a novel structure that was almost analogous to the familiar proton-ordered C1 hydrate but varied from the C1 hydrate in the orientations of water molecules.
The researchers demonstrated that proton disorder should take place at room temperature, thereby describing the X-ray diffraction and Raman spectrum data achieved in the experiment.
Journal Reference:
Wang, Y., et al. (2020) Novel Hydrogen Clathrate Hydrate. Physical Review Letters. doi.org/10.1103/PhysRevLett.125.255702.