In this interview, industry expert Michael Junglas discusses how PTFE and ETFE fluoropolymer films support reliable pharmaceutical packaging. The conversation explores their role in stoppers, plungers, and container closure systems, highlighting performance, drug compatibility, sterilization resistance, and material selection considerations for maintaining product stability and regulatory compliance.
What makes Norgard® PTFE and ETFE films appropriate for medical and pharmaceutical applications?
Norgard’s films were designed specifically for pharmaceutical interaction surfaces. They provide great purity, chemical inertness, and cleanliness, reducing interactions with hostile media and sensitive medication formulations.
They can endure sterilization and high temperatures while maintaining dimensional and mechanical stability, promoting long-term safety and reliability in medical systems.
Skived virgin PTFE films are externally evaluated for FDA and (EG) No. 1935/2004 certification, and the company provides regulatory alignment and paperwork for all applications.
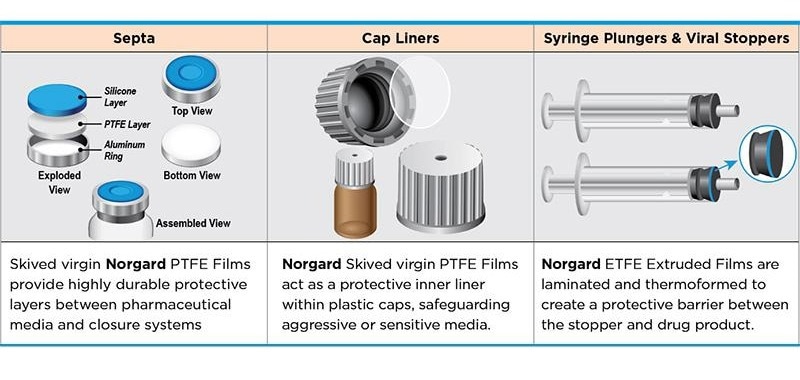
Image Credit: Saint-Gobain Tape Solutions
How does Norgard PTFE film increase closure performance and sealing integrity?
The skived PTFE layer ensures a stable, smooth, and chemically resistant surface that allows for a tight, dependable seal even when in contact with hostile pharmaceutical media.
It protects the cap's inside surface from degrading or softening and ensures that seal performance remains consistent throughout storage, which contributes to long-term container integrity.
Are Norgard films compatible with sterilizing procedures?
Yes. PTFE and ETFE are ideal for pharmaceutical sterilization due to their high temperature resistance and chemical stability. They maintain mechanical and barrier qualities during sterilizing and high-temperature filling operations.
Do these films maintain their performance after filling and storage?
They do. Their chemical resistance, thermal stability, and mechanical robustness ensure consistent performance from fill-finish to long-term storage, preserving seal integrity and providing barrier protection for sensitive media.
Can film characteristics be tailored for specific applications or production processes?
Absolutely. The company can provide customized thicknesses, widths, textures, surface treatments, and multilayer constructions.
Its skills include skiving, extrusion, surface treatments to make films bondable, laminating, and texturing. This allows Norgard to customize the film to fit specific sealing systems, plunger geometries, or customer processing needs.
Are PFAS-free alternatives available?
Yes. In addition to PTFE and ETFE, Norgard can provide PFAS-free solutions such as PMP films based on the application requirements.
These may be useful for applications that demand a PFAS-free alternative with great temperature and chemical resistance, high optical clarity, low moisture absorption, thermal stability, and high gas permeability/breathability.
Collaborating with its customers, Norgard produces PFAS-free solutions that meet regulatory requirements and customer sustainability goals.
How do Norgard films promote long-term medication stability?
Norgard’s films protect medicines from direct contact with elastomeric or polymeric components, lowering the danger of extractables, adsorption, degradation, and contamination. Their inert, ultra-clean surfaces and superior chemical resistance ensure the long-term stability of sensitive formulations such as biologics and vaccines.

Image Credit: Saint-Gobain Tape Solutions
Can Saint Gobain help with global supply and regulatory compliance?
Yes. Saint Gobain has a global production network that follows consistent quality processes, medical-grade documentation, and regulatory compliance. The company supports pharmaceutical OEMs worldwide, ensuring consistent supply and product consistency across geographies, making it a dependable long-term partner.
Why is ETFE used to cover syringe plungers rather than leaving the rubber exposed?
ETFE provides a chemically inert barrier between the rubber plunger and the drug formulation, preventing direct contact. This considerably minimizes extractables, particles, and contamination risk, resulting in a cleaner and more stable environment than uncoated rubber, which is especially critical for biologics and vaccines.
How does ETFE increase syringe plunger performance compared to uncoated rubber?
ETFE's low-friction surface ensures smooth plunger glide and uniform dose distribution, while its barrier function reduces medication interactions. This improves dosing performance and ensures compatibility with sensitive formulations over time.
Is the ETFE layer durable during deep drawing or thermoforming of the plunger?
Yes. Norgard ETFE extruded films have outstanding thermoformability, allowing for wrinkle-free, secure lamination even in complex plunger geometries. They can withstand deep pulling without rupturing, ensuring both mechanical integrity and barrier performance.
Can ETFE-laminated plungers handle high-speed automation?
Yes. The films' thermoformability and surface properties enable high-speed automated lamination and plunger manufacture, ensuring quality and consistency at scale.
What are the advantages of these products in comparison to other options on the market?
Saint Gobain offers high-purity ETFE films, superior extrusion technology, and global quality standards. These films provide superior thermoforming performance for demanding shapes, surface polish, and barrier qualities.
The company offers bespoke designs, strong technical support, and stable worldwide supply to ensure consistent and regulatory-aligned performance.
What impact does the PTFE liner have on product stability over time?
PTFE's non-reactive, impermeable, and solvent-resistant properties ensure that the formulation and cap materials do not interact throughout a product’s shelf life. This guards against chemical degradation, maintains purity, and allows for reliable long-term storage, making it perfect for high-value or delicate pharmaceutical items.
How does a PTFE cap liner affect closure integrity over time?
The skived PTFE layer creates a stable and chemically resistant surface, ensuring a tight seal even with aggressive media. It contributes to the maintenance of closure performance and container integrity during storage.
How can PTFE film enhance closure performance and sealing integrity?
The ultra-smooth surface prevents particle formation and extractables, while maintaining a clean sealing surface. Its mechanical stability and thermoformability provide exact fit and consistent seal compression, ensuring container closure integrity.
What types of pharmaceutical drugs usually require PTFE cap liners?
Bottles carrying harsh solvents, delicate vaccine components, diagnostic reagents, and other media where even minor contact with polymers could jeopardize safety or stability. PTFE is required anywhere extractables, leachables, or chemical incompatibility are a concern.
Can PTFE sheets be thermoformed into cap liner shapes with no defects?
Yes. Skived virgin PTFE films, created with high-quality resins and best-in-class production, may be thermoformed into cap liner geometries without ruptures or wrinkles, allowing for stable insertion and positioning.
Can PTFE film be tailored to specific cap designs and manufacturing processes?
Saint Gobain offers custom thicknesses and widths with precise tolerances, surface treatments, and multi-layer choices as needed. Its skiving technology provides exact thickness control across the whole length and width of the film, resulting in smooth, defect-free surfaces matched to unique filling or capping processed.
Why use Norgard Skived PTFE Film?
Norgard skived virgin PTFE films have extraordinarily high fluoropolymer purity, resulting in limited extractables and strong solvent resistance, helping to safeguard medication stability more consistently than typical alternatives. Precision skiving, consistent thickness control, and outstanding thermoformability ensure defect-free forming and easy assembly.
Why should PTFE-laminated septa be used instead of standard rubber septa?
Standard rubber can react with pharmaceutical media, release extractables, and deteriorate in harsh solvents. Laminating rubber with virgin PTFE forms an ultra-clean, impermeable, and chemically inert barrier that protects sensitive formulations, resulting in improved purity and more stable long-term performance.
What unique performance improvements does PTFE provide for septa systems?
PTFE provides a non-reactive barrier that avoids chemical interaction with the medication, has great pierceability, resists aggressive solvents and biologics, and significantly enhances cleanliness and lifespan, which is especially useful in high-frequency vial access conditions.
Will PTFE laminations impair pierceability for septa applications?
No. The PTFE-to-rubber multilayer is intended to maintain pierceability while providing a clean, impenetrable barrier. It ensures consistent, dependable access while protecting against interaction-related contamination.
How does Saint Gobain maintain the cleanliness and purity of its PTFE-laminated septa films?
Its skived virgin PTFE films have exceptionally high fluoropolymer purity, hyper-smooth surfaces, and extremely little particle production. When laminated to the elastomer, the multilayer design creates a very dependable contamination barrier that meets pharmaceutical and diagnostic standards.
Why use Saint Gobain's product over the competition?
Exceptional polymer purity and surface quality result in low extractables and high chemical resistance. Its best-in-class etching makes the film bondable, allowing for strong adhesion to rubber, and precision skiving provides consistent thickness and defect-free surfaces, resulting in robust, clean, and durable septa.
How do advanced fluoropolymer barrier materials, such as PTFE and ETFE, contribute to the performance, safety, and regulatory compliance of modern drug delivery and container sealing systems?
In important drug delivery and container sealing systems, the interface is crucial. Manufacturers can reduce extractables and particulates, maintain seal integrity, and support long-term stability for sensitive formulations by incorporating inert, ultra-clean fluoropolymer barriers exactly where the drug meets the component, such as Norgard PTFE for cap liners and septa and Norgard ETFE for syringe plungers.
These films are designed to withstand sterilization, high-speed automation, and lengthy storage while adhering to global regulatory demands and quality standards.
When PFAS-free materials are required, the Tape Solutions team has the expertise to provide co-development support to help satisfy both technical and sustainability objectives.
The end result is a cleaner, safer, and more reliable path from fill-to-patient, supported by a partner with the materials expertise and manufacturing depth to deliver at scale.
About Michael Junglas 
Michael Junglas holds a Dipl.-Ing. in Mechanical Engineering with a specialization in Mechatronics from Hochschule Bonn-Rhein-Sieg and an M.Sc. in Economics from RWTH Aachen University. At Saint-Gobain, he has held multiple product management and market leadership roles focused on specialty films and application engineering.

This information has been sourced, reviewed and adapted from materials provided by Saint Gobain PPL Corp.
For more information on this source, please visit Saint Gobain PPL Corp.
Disclaimer: The views expressed here are those of the interviewee and do not necessarily represent the views of AZoM.com Limited (T/A) AZoNetwork, the owner and operator of this website. This disclaimer forms part of the Terms and Conditions of use of this website.