The MicroCal PEAQ-DSC Automated system is designed for gold standard protein stability analysis for a regulated environment. The system is an automated, integrated platform that delivers high throughput, highly sensitive protein stability analysis with low sample consumption for improved productivity. All cell filling and cleaning operations are 100% automated, allowing walk-away operation.
Data generated by the MicroCal PEAQ-DSC Automated provide vital guidance for biopharmaceutical development in protein engineering, manufacturing change control, process development, (pre)formulation development, and biocomparability and biosimilarity studies.
It is possible to have 24-hour screening with unattended operation, while the integrated software streamlines workflows, enables non-subjective data analysis, performance qualification and compliance with 21 CFR Part 11 and Annex 11 regulations, which combine to deliver high integrity data and drive productivity in biopharmaceutical research.
Differential Scanning Calorimetry (DSC) is a strong analytical tool designed to perform characterization of the thermal stability of proteins and other biomolecules. The method measures the temperature (TM) and enthalpy (ΔH) of thermally-induced structural transitions of molecules in solution.
This information provides valuable insights into factors that stabilize or destabilize proteins, micellar complexes, nucleic acids, and other macromolecular systems.
The data is used to estimate the shelf-life of biomolecular products including biopharmaceuticals to develop purification strategies, to enable batch-to-batch and biosimilar versus innovator molecule comparisons, to rank the affinities of ligands to their protein targets in small molecule drug discovery programs, and to characterize and evaluate protein constructs.
The MicroCal PEAQ-DSC Automated system features software that streamlines workflows through simplified experimental arrangement and flexible instrument scheduling. Automated data analysis supports the production of high integrity thermal stability data and matches all regulatory requirements, simultaneously allowing easy integration into current data handling and transfer systems.
Key Features
The key features of the MicroCal PEAQ-DSC Automated System are given below:
- Gold standard stability-indicating method requiring minimum assay development
- Direct and label-free measurement of biomolecular native-state stability in solution
- Standard 96-well plate format for easy loading and high capacity, with thermostatically-controlled storage of up to six plates
- Fully integrated autosampler enables unattended analysis of up to 50 samples per day
- Automated cell filling and cell cleaning functions allow unattended operation
- Measurement of extremely tight binding constants, up to 1020M-1
- Powerful MicroCal PEAQ-DSC software reduces typical data analysis time and includes:
- PEAQ-Performance: Automatically detects and validates system readiness for optimal performance
- PEAQ-Compliance: The system is available with Malvern Panalytical Access Controller (MAC) to limit access to user-defined SOPs and data analysis features. Data can be displayed using the Report Generator with user details and the ability to sign using electronic signatures to allow the integration of the workflow into the company’s quality system and help compliance with 21 CFR Part 11 and Annex 11 regulations. This is available as an optional accessory
- PEAQ-Compare: For the quantitative comparison of DSC traces for comparability studies, suitable for biosimilarity and batch-to-batch studies
- PEAQ-Smart (including PEAQ-Finder): For SOP-based operation and data analysis. Offers new algorithms to detect even subtle peaks and shoulders, enabling the automatic, non-subjective identification of multiple transitions, such as those observed in multi- domain proteins
- Network-ready: email updates sent during the analysis to keep users informed of assay progress
The MicroCal PEAQ-DSC is also available as a manual system without the autosampler.
How It Works
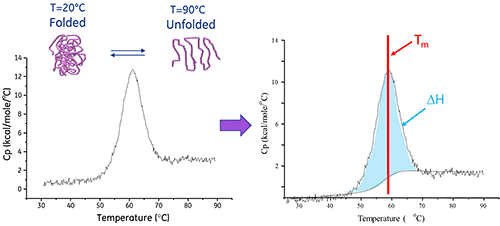
Functional structures created using proteins and other macromolecules frequently undergo temperature-induced conformational changes, such as unfolding. These changes cause heat absorption as a result of the redistribution of non-covalent bonds within the molecule. Differential Scanning Calorimeters calculate this heat uptake highly precisely.
The thermal core of the MicroCal PEAQ-DSC Automated system has a reference cell (containing a matched buffer solution) and a sample cell (containing the sample of interest), both of which are placed within an insulating jacket. These two cells are constantly maintained at the same temperature, and while performing a measurement, they are heated at a constant scan rate.
When the molecule within the sample cell unfolds, heat is absorbed, creating a temperature difference (ΔT) between the reference cell and the sample cell. This results in a thermal gradient across the Peltier units, generating a proportional voltage, which is converted to power to form a feedback loop to the Peltier units, in order to return ΔT to zero.
As protein unfolding is an endothermic event, it is seen as a positive displacement in the thermogram. This protein ‘melting’ transition’s midpoint is the TM, and the area under the curve is the enthalpy (ΔH) of the unfolding process (see image below).
Specifications
General
- Technology: Differential Scanning Calorimetry
- Measurement type: Temperature midpoint (TM); Enthalpy (ΔH); Heat capacity change (ΔCp)
Cell
- Cell: Capillary
- Cell material: Tantalum
- Cell volume: 130 µL
Sample
- Sample capacity: 288 (6 × 96-well plates)
- Sample volume: 325 µL
- Typical sample concentration: 0.01 mg/mL – 10 mg/mL 1
- Sample throughput: ≤50 samples/day
- Sample storage temperature range: 4 °C – 40 °C
System
- Noise: 0.05 µCal/°C 2
- Baseline repeatability: 1 µCal/°C 2
- Response time: 5s 2
- Repeatability: <0.2 µCal/°C 3
- Reproducibility: <0.08 °C St. Dev. TM and <2% RSD on ΔH 4
- System reproducibility: <0.1°C St. Dev. TM and <5% RSD on ΔH 4
- Multiple feedback modes: Yes (passive, high gain and low gain)
- Temperature range: 2 °C to 130 °C 2, 5
- Maximum scan rate: 240 °C/h
- Reverse scanning: Yes
- Pressure perturbation calorimetry (PPC): N/A
- Cleaning solvents: Water and detergent used as standard
- Cleaning routines: Three pre-programmed routines
Software
- 21 CFR part 11: Yes, with PEAQ-Compliance software option
- Network ready: Yes, with email alert capability
Operating Environment
- Operating temperature (°C): +10 °C to +28 °C
- Storage temperature: -20 °C to +50 °C
- Humidity: 10% to 70%, non-condensing (10% to 90% for storage)
- Ingress Protection (IP) rating: IP21
- Power: 100-240 V A/C, 50/60 Hz, 70 W (cell), 400 W (max, autosampler), PC as supplied
- Certification: CE (EN61010-1), EN61326-1, FCC, ICES, VCCI), EMC (EN61326-2-1, ISO9001:2008
Weight and Dimensions
- Dimensions (W, D, H): 101 cm × 68 cm × 70 cm
- Weight: Approx. 25 kg
Notes
1 Sample dependent
2 Typical results for ribonuclease (RNase) in 50 mM KAc buffer at pH 5.5, at 60 °C/h with passive feedback
3 Rescans of a stable buffer
4 Using ribonuclease (RNase)
5 Range may be extended down to -10 °C upon request